
99) One-Step Radical-Induced Synthesis of Graft Copolymers for Effective Compatibilization of Polyethylene and Polypropylene
Moritz Kranzlein, Shilin Cui, Jenny Hu, Anne M. LaPointe, Brett P. Fors, and Geoffrey W. Coates
J. Am. Chem. Soc. 2025. https://doi.org/10.1021/jacs.5c03869.

98) Controlled anionic polymerization mediated by carbon dioxide
Paige E. Jacky, Alexandra D. Easley, and Brett P. Fors
Nat. Chem. 2025. https://doi.org/10.1038/s41557-025-01819-7.

97) Designing Highly Conductive Anion Exchange Membranes: Tuning Domain Continuity with ABC Block Copolymer Self-Assembly
Alexandra J. Macbeth, Danielle Markovich, Abigail I. Taylor, Ethan B. Flanagan, Julia E. Borowski, Jesse H. Hsu, Cheyenne R. Peltier, David A. Muller, Brett P. Fors, Kevin J. T. Noonan, and Geoffrey W. Coates
J. Am. Chem. Soc. 2025, https://doi.org/10.1021/jacs.5c03175.

96) Radical Deamination of Primary Amines for Initiation of Controlled Polymerization
Megan E. Driscoll, Bryce T. Nicholls, and Brett P. Fors
J. Am. Chem. Soc. 2025, https://doi.org/10.1021/jacs.5c02493.

95) Designing Polymers with Molecular Weight Distribution-Based Machine Learning
Jenny Hu, Zachary M. Sparrow, Brian G. Ernst, Spencer M. Mattes, Geoffrey W. Coates, Robert A. DiStasio Jr., and Brett P. Fors
J. Am. Chem. Soc. 2025, https://doi.org/10.1021/jacs.4c16325.
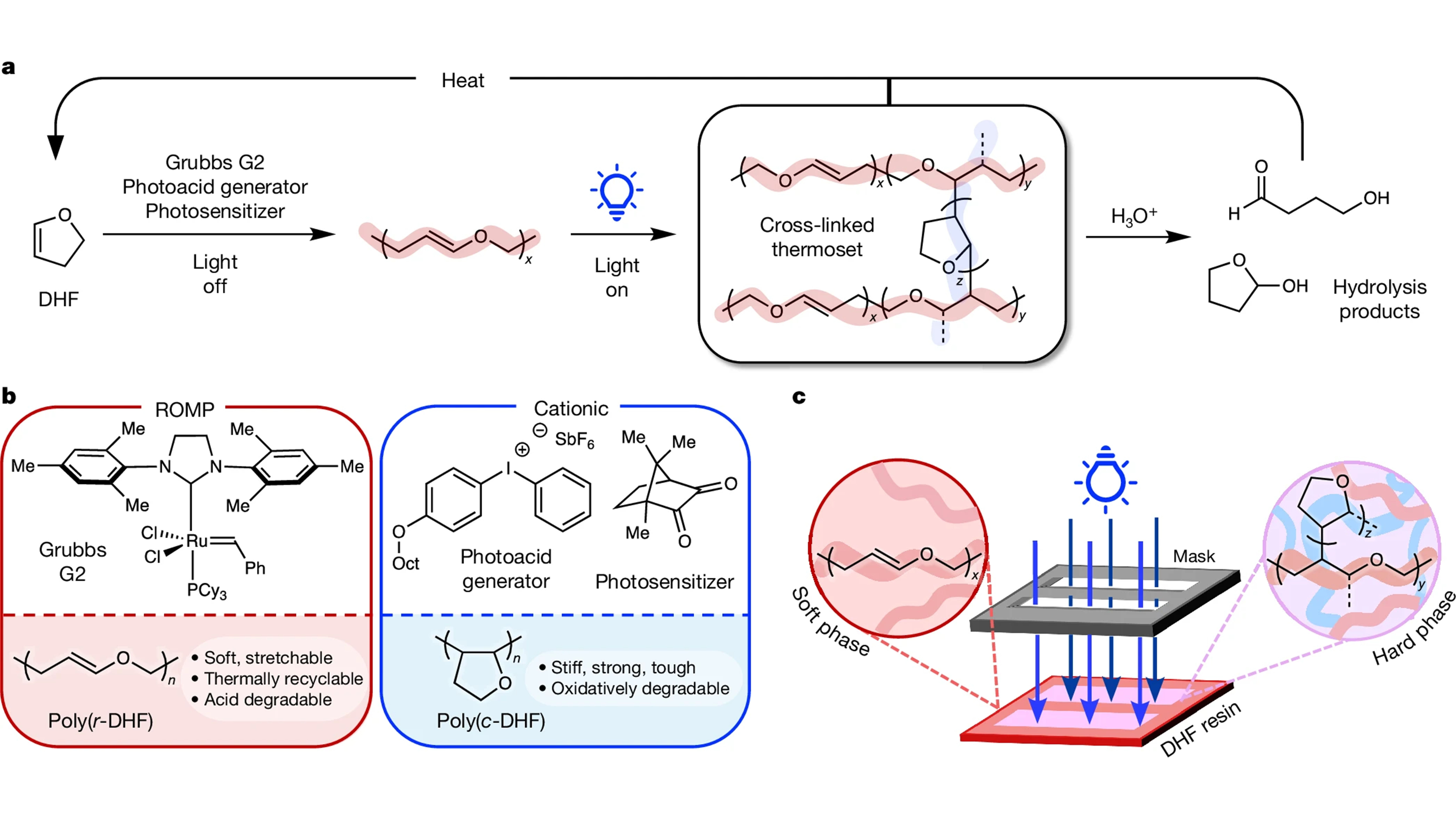
94) Degradable Thermosets via orthogonal polymerizations of a single monomer
Reagan J. Dreiling, Kathleen Huynh, and Brett P. Fors
Nature 2025, 638, 120–125.

93) Multimaterial Thermoset Synthesis: Switching Polymerization Mechanism with Light Dosage
Yuting Ma,‡ Reagan J. Dreiling,‡ Elizabeth A. Recker, Ji-Won Kim, Shelby L. Shankel, Jenny Hu, Alexandra D. Easley, Zachariah A. Page, Tristan H. Lambert, and Brett P. Fors
ACS Cent. Sci. 2024, Accepted Article.

92) Hydrazine-Catalysed Ring–Opening Metathesis Polymerization Of Cyclobutenes
Julian S. Kellner-Rogers, Jesse H. Hsu, Ivan Keresztes, Brett P. Fors, Tristan Hayes Lambert
Angew. Chem. Int. Ed. 2024, e202413093.

91) Low-Dose Mapping of Ionic Channel Architectures in Fuel-Cell Polymer Membranes with Cryo-STEM-EELS
Danielle Markovich, Jesse H. Hsu, Brett P. Fors, David A. Muller, and Lena F. Kourkoutis
Microsc. Microanal. 2024, 30, 1743-1744.

90) Direct Functionalization of Polyethylene Surfaces with High-Density Polymer Brushes
Anna E. Ringuette, Gozde Aktas Eken, Amaya B. Garnenez, Adriana I. Palmieri, Christopher K. Ober, Geoffrey W. Coates and Brett P. Fors
J. Am. Chem. Soc. 2024, 146, 30, 20563–20568.

89) Closing the Loop on Thermoset Plastic Recycling, Perspective
Bryce T. Nicholls and Brett P. Fors
Science. 2024, 384, 6692, 156-157.
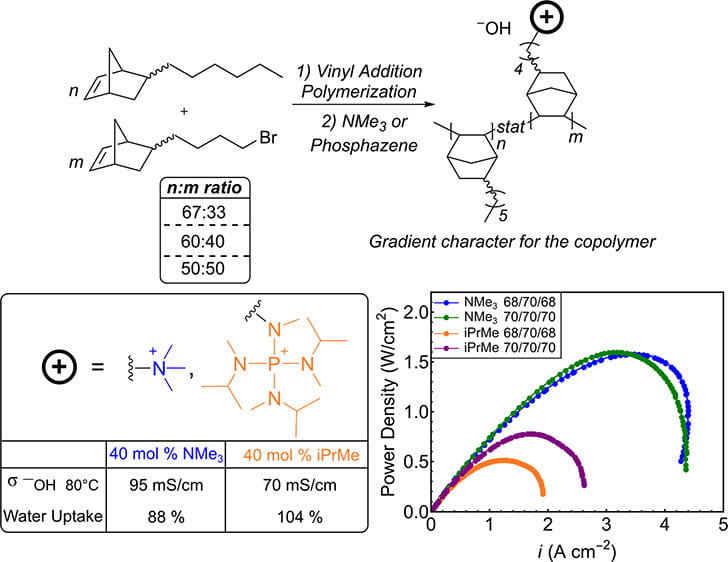
88) Comparing Ammonium and Tetraaminophosphonium Anion-Exchange Membranes Derived from Vinyl-Addition Polynorbornene Copolymers
Jamie C. Gaitor, Ami C. Yang-Neyerlin, Daneille Markovich, Brett P. Fors, Geoffrey W. Coates, Lena F. Kourkoutis, Bryan S. Pivovar, Tomasz Kowalewski, and Kevin J. T. Noonan
ACS Appl. Energy Mater. 2024, 7, 4, 1517–1526.
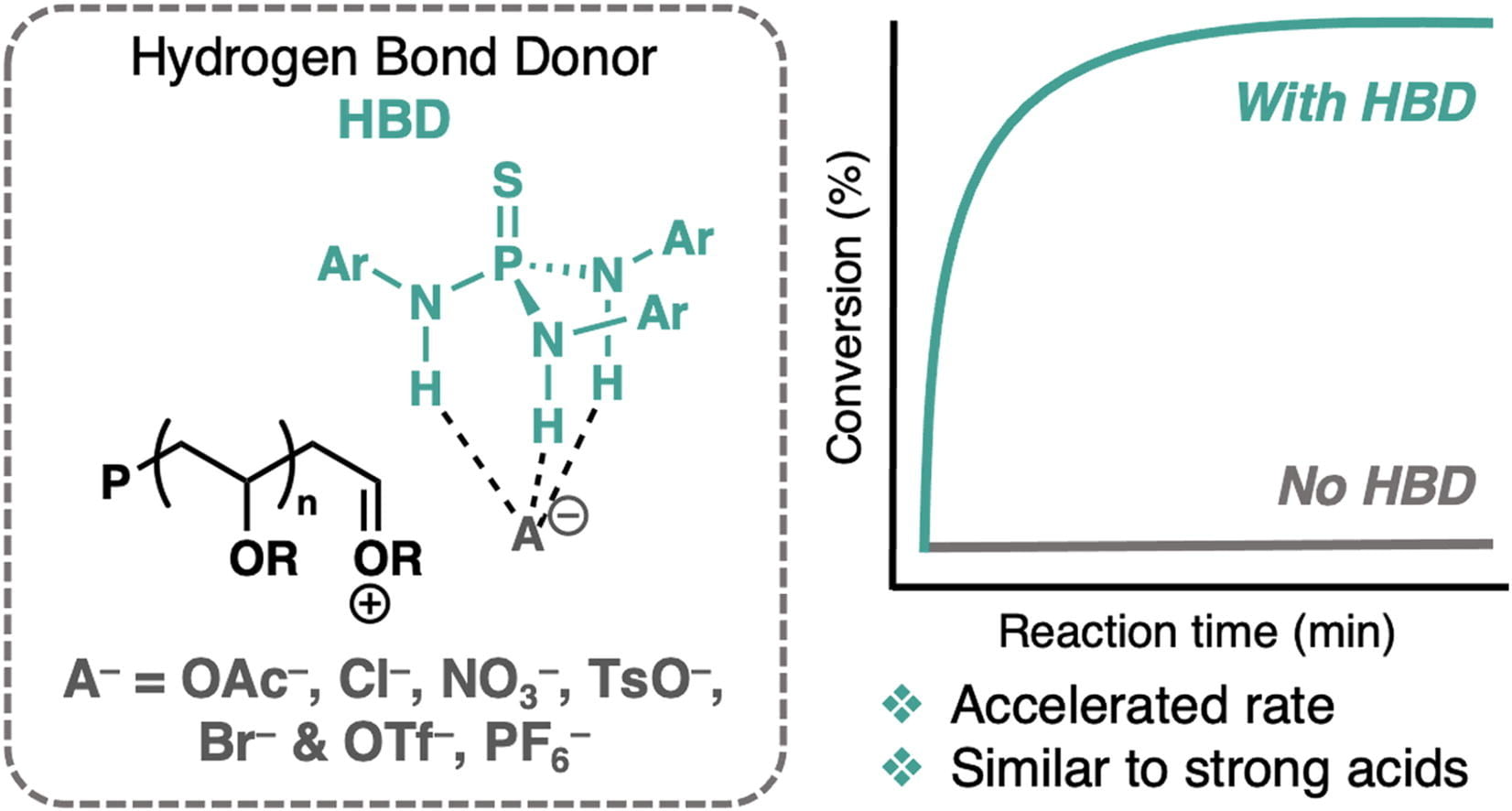
87) Accelerating Cationic Polymerizations with a Hydrogen Bond Donor
Shelby L. Shankel,‡ Yuting Ma,‡ Jesse A. Spivey , Leila Filien, Tristan H. Lambert, Brett P. Fors
Eur. Polym. J. 2024, 207, 112814.
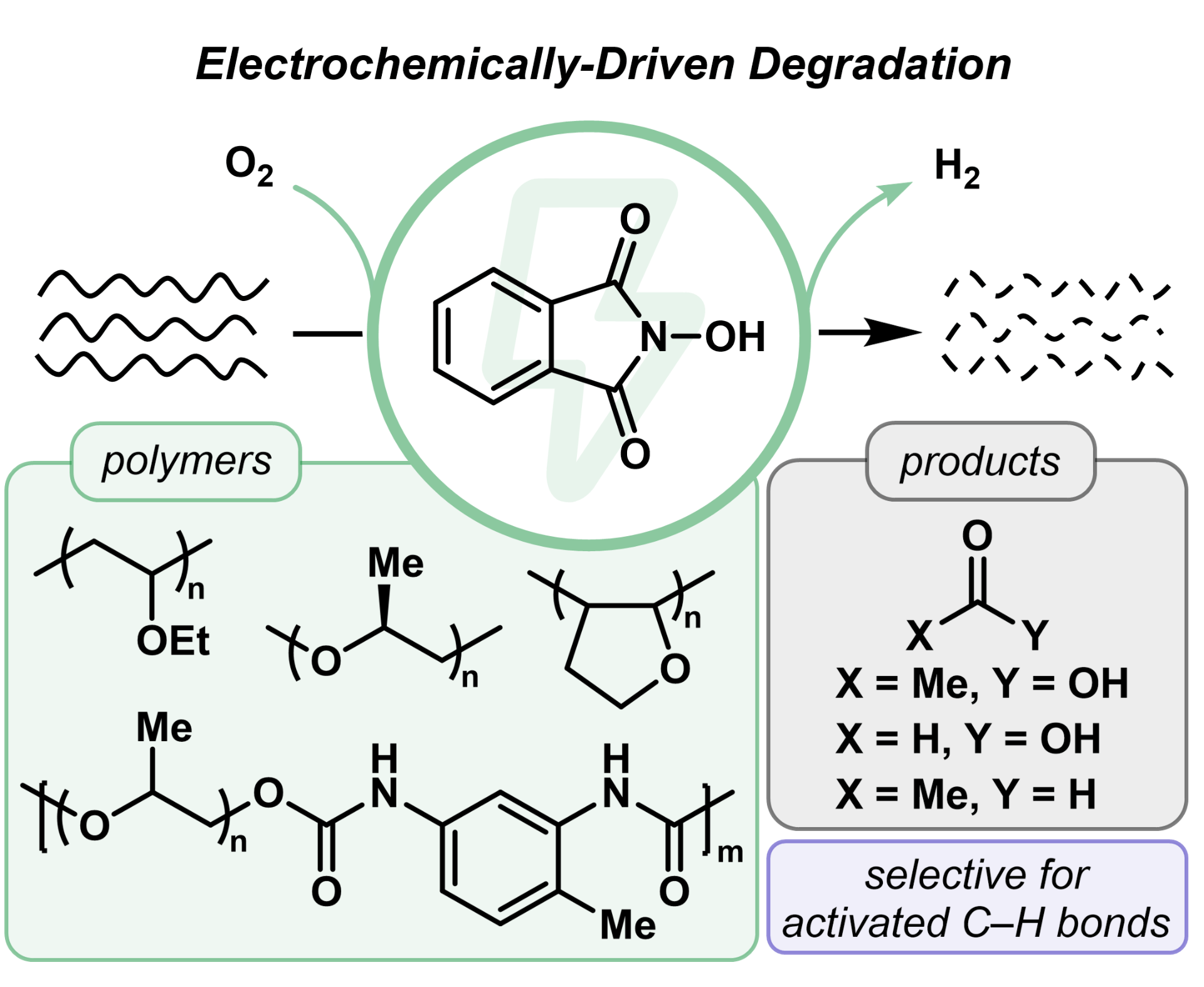
86) Selective Electrocatalytic Degradation of Ether-Containing Polymers
Jesse H. Hsu, Tyler E. Ball,‡ Sewon Oh,‡ Erin E. Stache, and Brett P. Fors
Angew. Chem. Int. Ed. 2024, 63, e2023165.
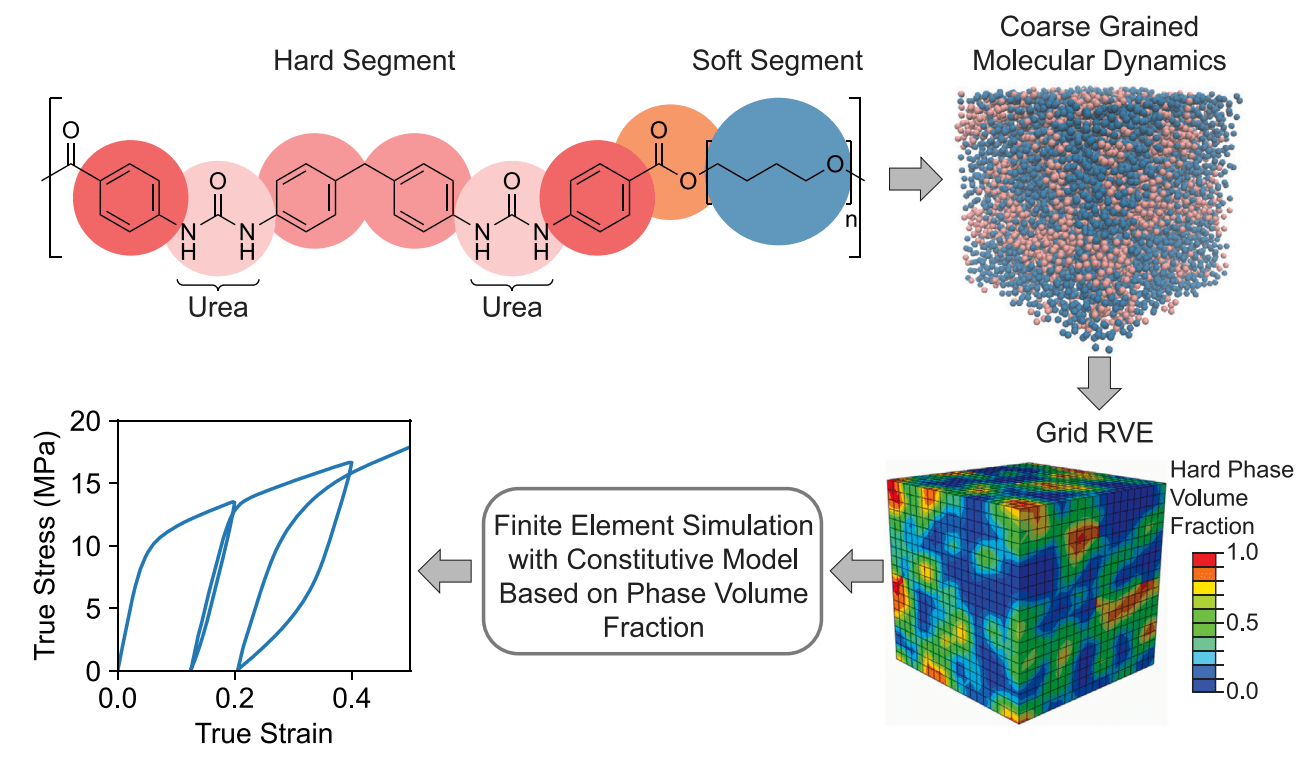
85) Elucidating the Impact of Microstructure on Mechanical Properties of Phase-Segregated Polyurea: Finite Element Modeling of Molecular Dynamics Derived Microstructures
Steven J. Yang, Stephanie I. Rosenbloom, Brett P. Fors, and Meredith N. Silberstein
Mech. Mater. 2024, 188, 104863.

84) Revealing the Internal Architecture of Alkaline Fuel Cell Membranes with Cryo-4D-STEM and Cryo-STEM-EELS
Danielle Markovich, Michael Colletta, Yue Yu, Megan Treichel, Jesse H. Hsu, Bryan Pivovar, Brett P. Fors, Kevin J. T. Noonan, and Lena F. Kourkoutis
Microsc. Microanal. 2023, 29, 1274.
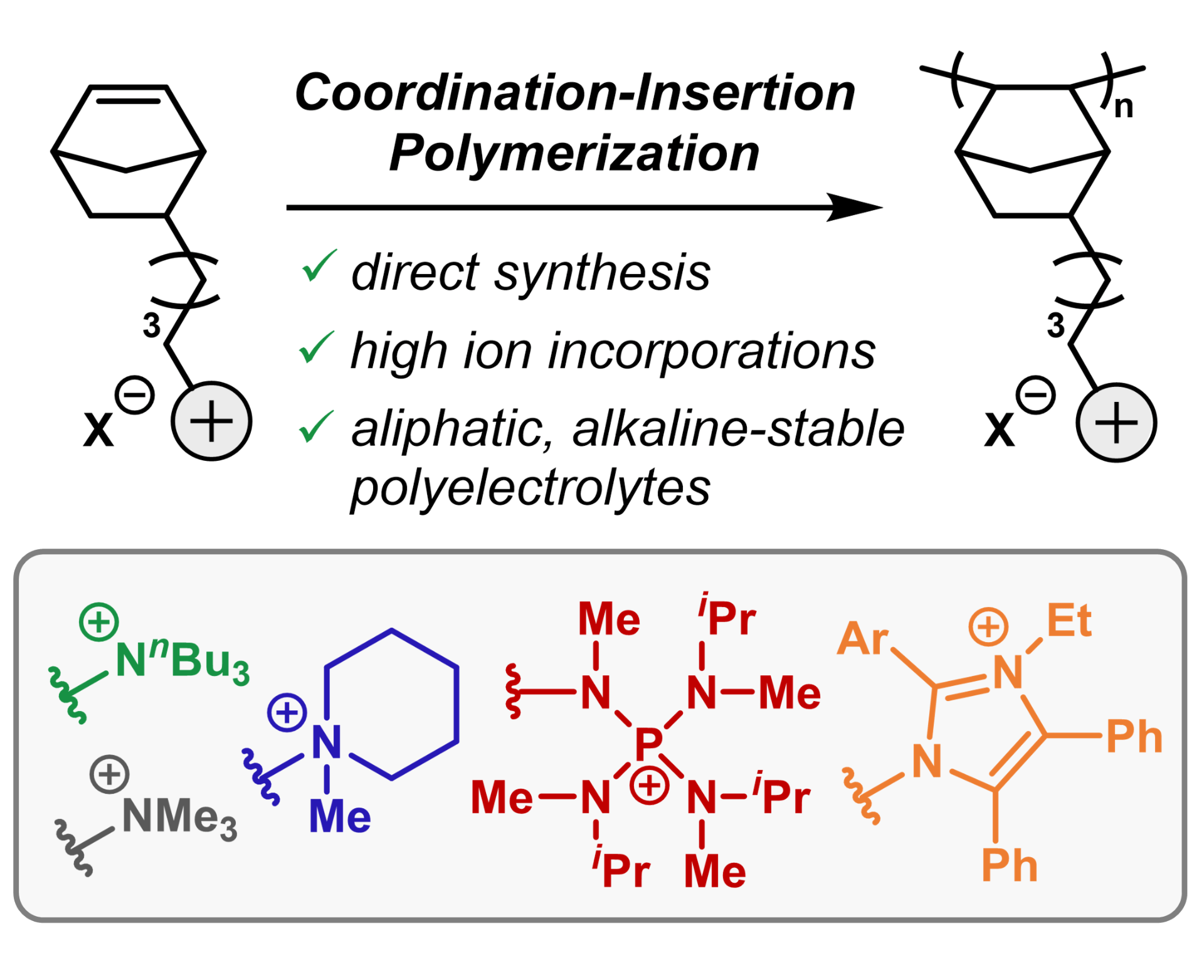
83) Direct Insertion Polymerization of Ionic Monomers: Rapid Production of Anion Exchange Membranes
Jesse H. Hsu, Cheyenne R. Peltier, Megan Treichel, Jamie C. Gaitor, Qihao Li, Renee Girbau, Alexandra J. Macbeth, Héctor D. Abruña, Kevin J. T. Noonan, Geoffrey W. Coates, and Brett P. Fors
Angew. Chem. Int. Ed. 2023, 62, e202304778.
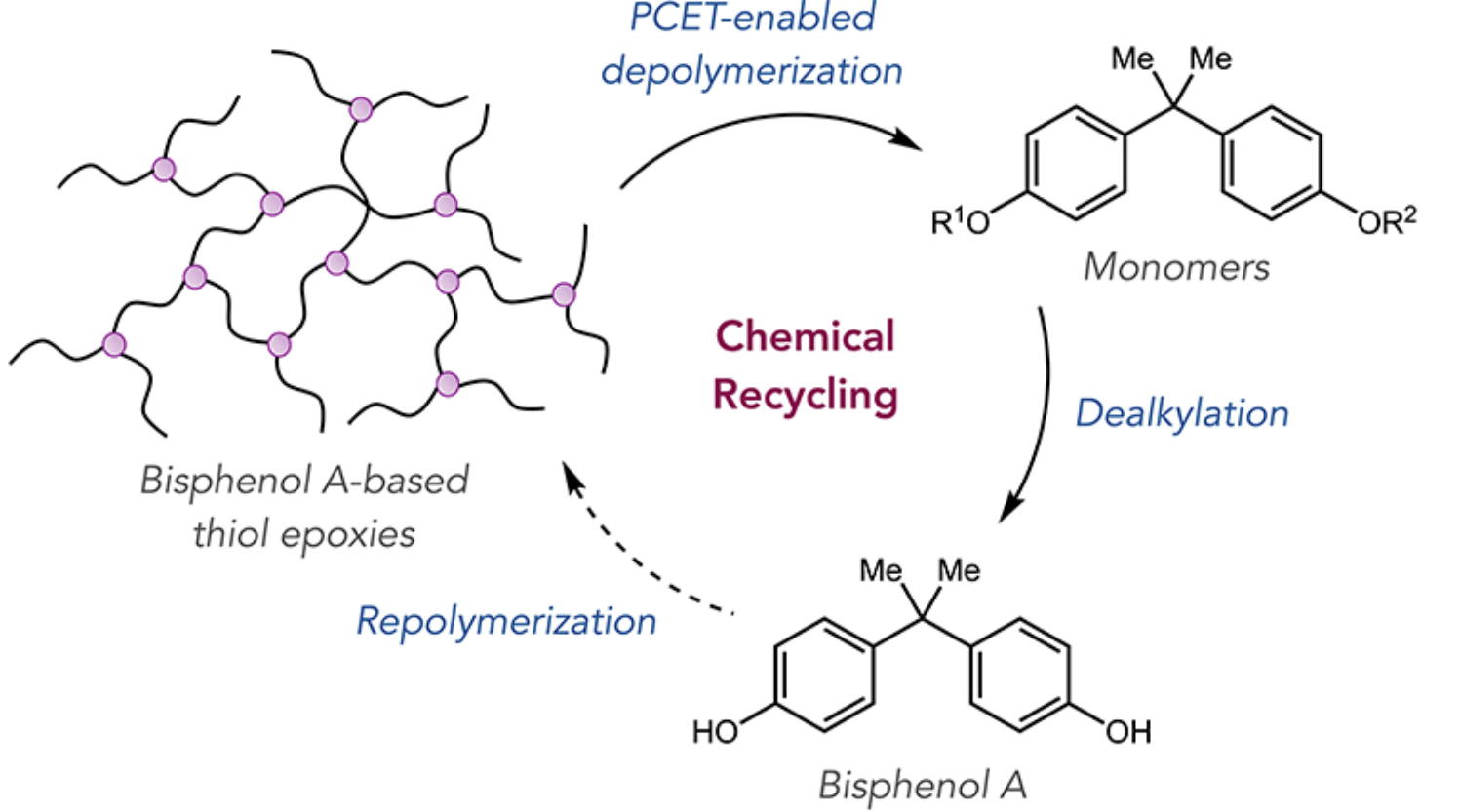
82) Chemical Recycling of Thiol Epoxy Thermosets via Light-Driven C-C Bond Cleavage
Suong T. Nguyen, Lydia R. Fries,‡ James H. Cox,‡ Yuting Ma, Brett P. Fors, and Robert R. Knowles
J. Am. Chem. Soc. 2023, 145, 11151.
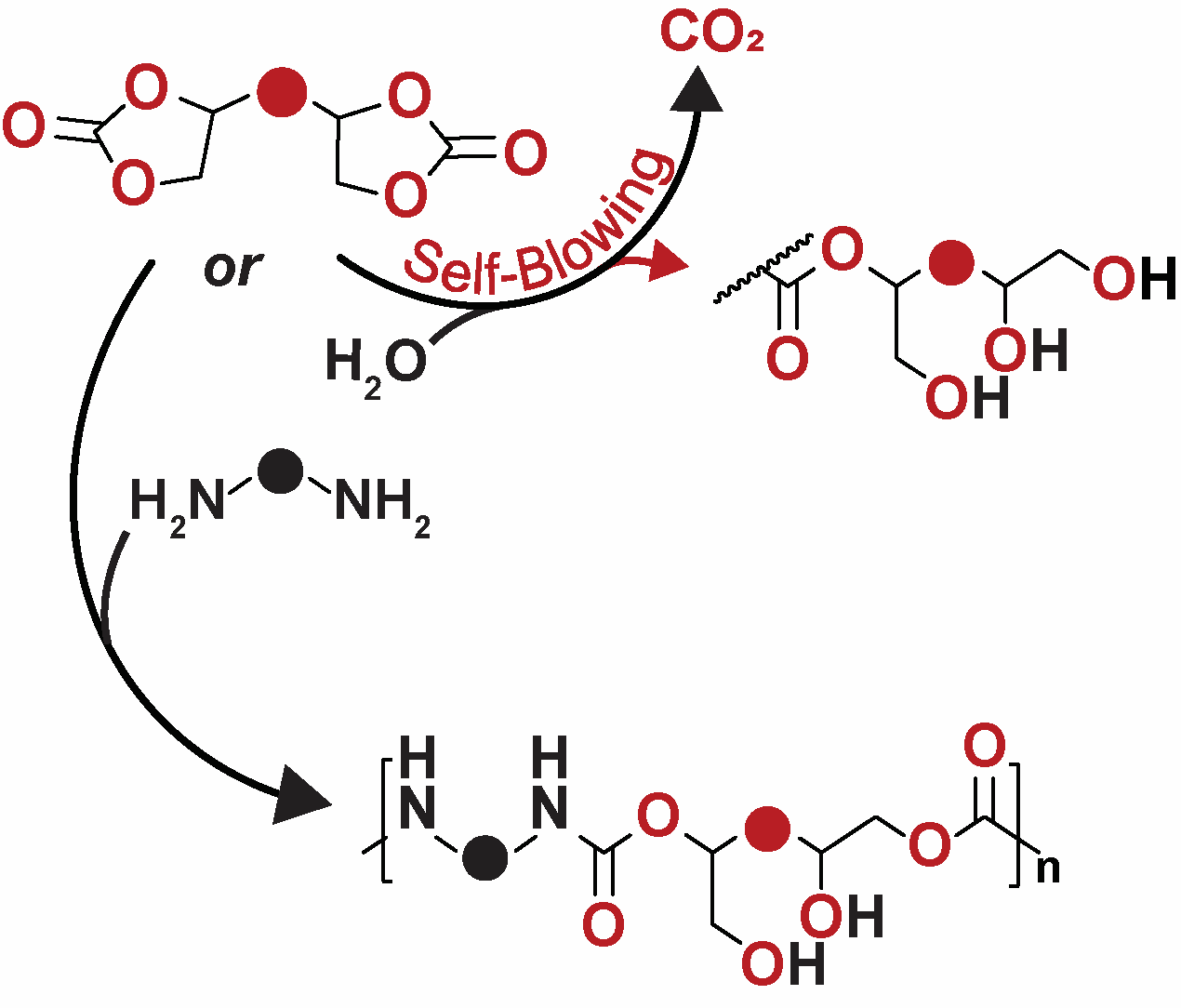
81) Safer Polyurethane Foams with Cyclic Carbonates, Highlight
Alexandra D. Easley, Alison Mangano, and Brett P. Fors
Angew. Chem. Int. Ed. 2023, 62, e202218062
80) Mixing Discrete Block Co-Oligomers: When Does It Behave like a Disperse Sample?
Brigitte A. G. Lamers, Brett P. Fors, and E. W. Meijer
J. Polym. Sci. 2022, 1.
79) Scalable, Green Chain Transfer Agent for Cationic RAFT Polymerizations
Scott W. Spring, Chloe S. Cerione,‡ Jesse H. Hsu,‡ Shelby L. Shankel, and Brett P. Fors
Chin. J. Chem. 2022, 41, 399.
78) Differential Impact of the COVID-19 Pandemic on Female Graduate Students and Postdocs in the Chemical Sciences
Renee J. Sifri, Elizabeth A. McLoughlin, Brett P. Fors, and Shima Salehi
J. Chem. Ed. 2022, 99, 3461.
77) Moisture Tolerant Cationic RAFT Polymerization of Vinyl Ethers
Shelby L. Shankel, Tristan H. Lambert, and Brett P. Fors
Polym. Chem. 2022, 13, 5979.
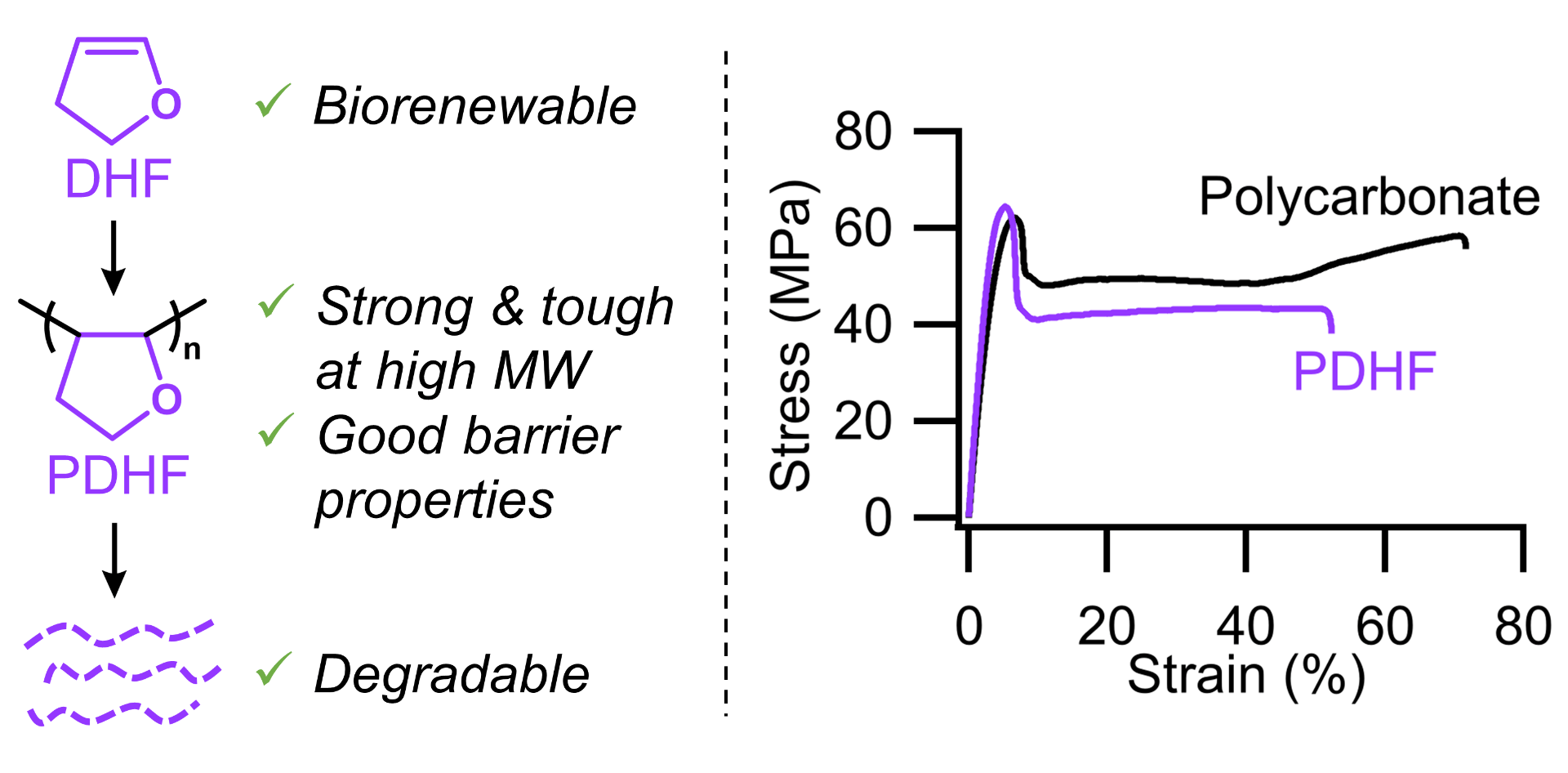
76) Poly(2,3-Dihydrofuran): A Strong, Biorenewable, and Degradable Thermoplastic Synthesized via Room Temperature Cationic Polymerization
Scott W. Spring, Jesse H. Hsu, Renee J. Sifri, Szu-Ming Yang, Chloe S. Cerione, Tristan H. Lambert, Christopher J. Ellison, and Brett P. Fors
J. Am. Chem. Soc. 2022, 144, 15727.
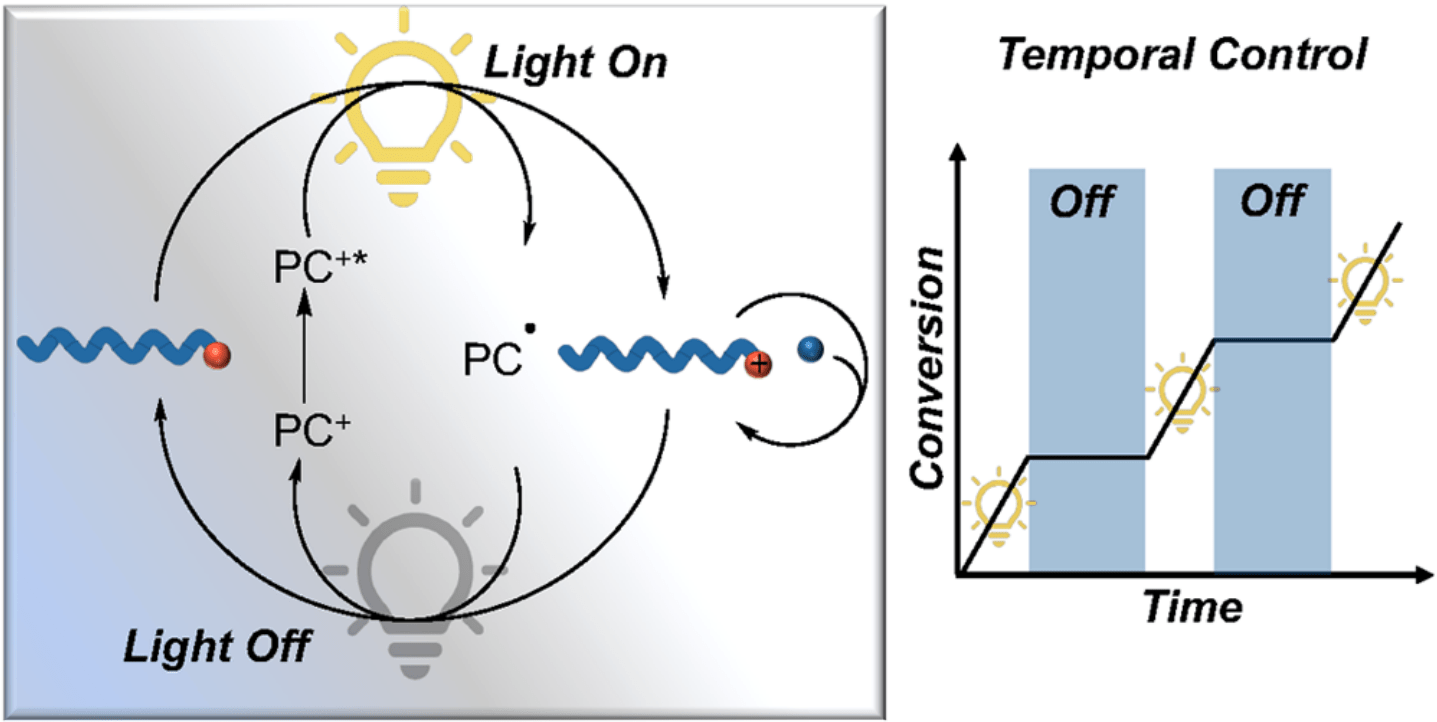
75) Photoredox Catalysis in Photocontrolled Cationic Polymerizations of Vinyl Ethers
Renee J. Sifri, Yuting Ma, and Brett P. Fors
Acc. Chem. Res. 2022, 14, 1960.
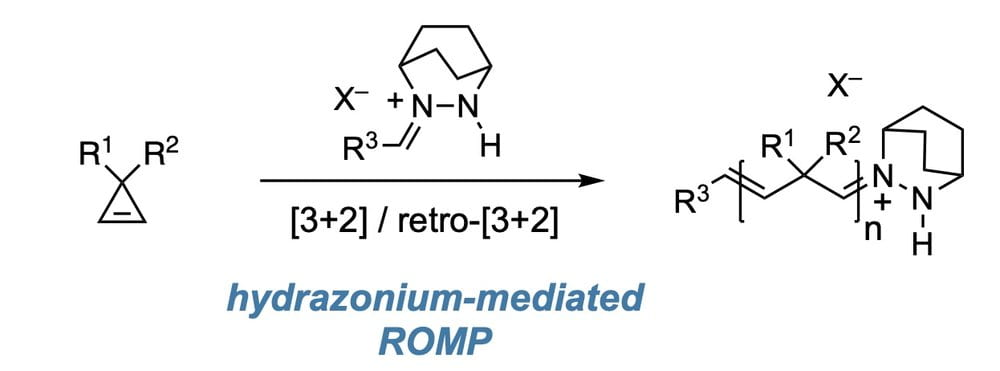
74) Metal–Free Ring–Opening Metathesis Polymerization with Hydrazonium Initiators
Phong K. Quach, Jesse H. Hsu, Ivan Keresztes, Brett P. Fors, and Tristan H. Lambert
Angew. Chem. Int. Ed. 2022, 61, e202203344.
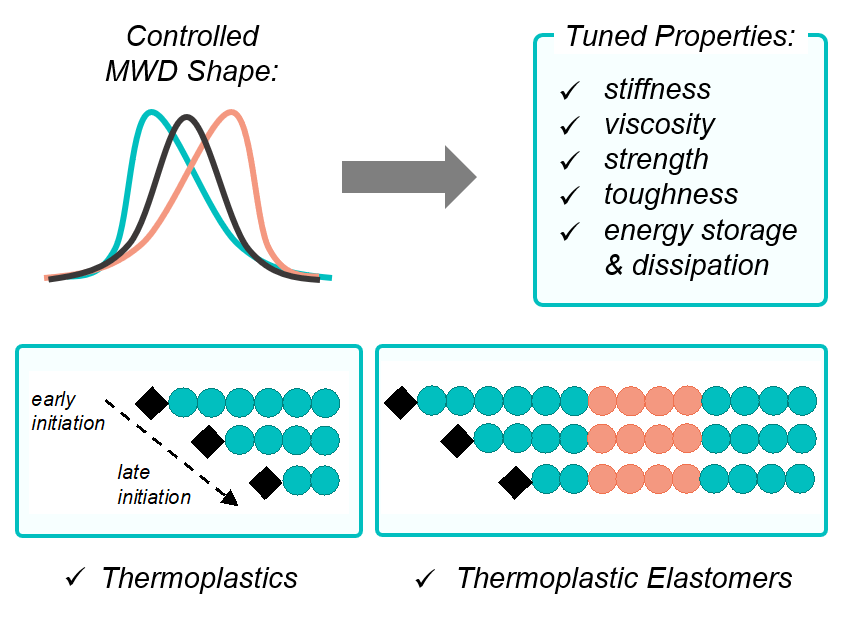
73) Controlling the Shape of the Molecular Weight Distribution for Tailored Tensile and Rheological Properties in Thermoplastics and Thermoplastic Elastomers
Stephanie I. Rosenbloom, Jesse H. Hsu, and Brett P. Fors
J. Polym. Sci. 2022, 60, 1291.
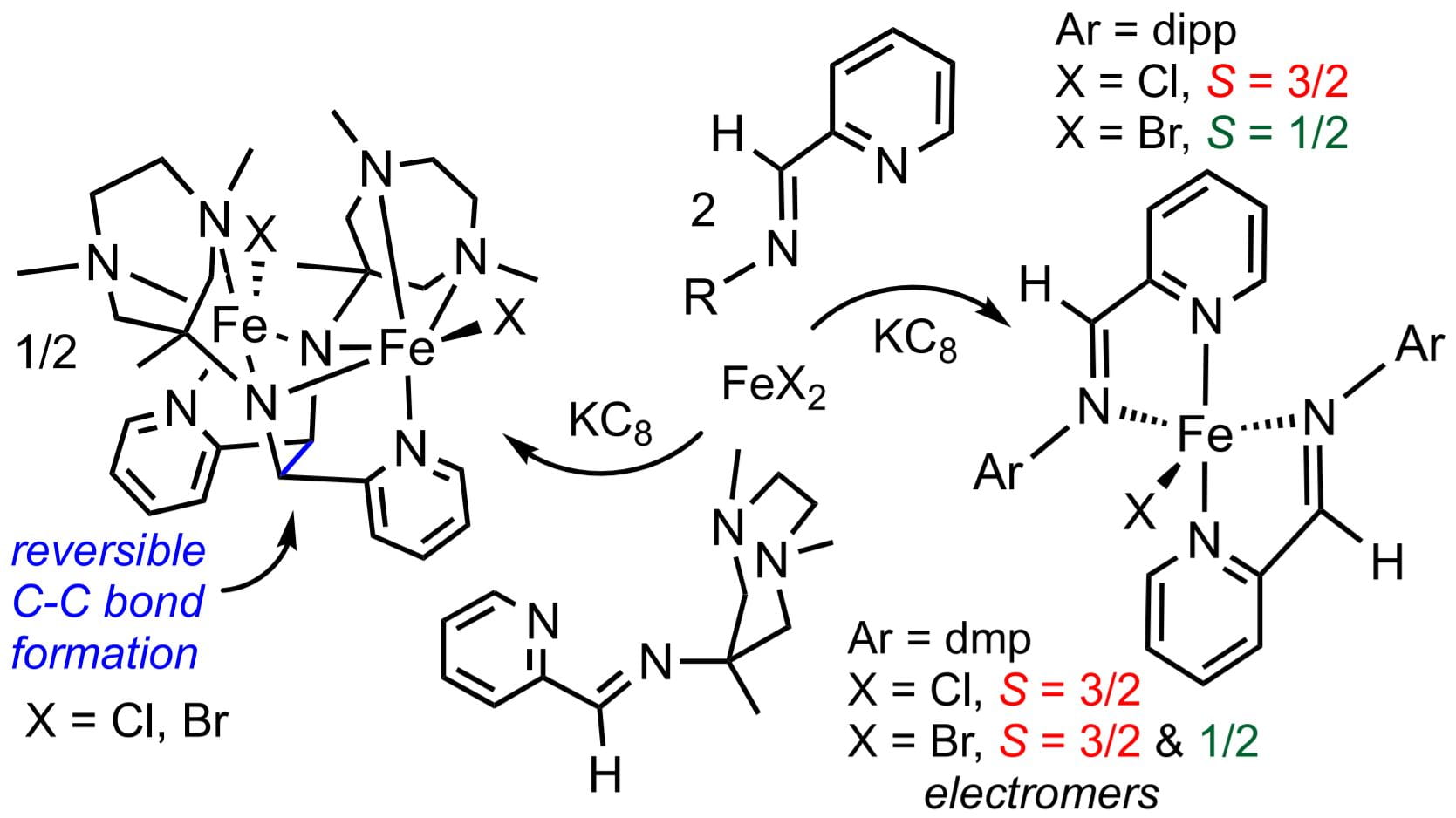
72) Reversible C–C Bond Formation, Halide Abstraction, and Electromers in Complexes of Iron Containing Redox-Noninnocent Pyridine-imine Ligands
Devika Pokhriyal, Spencer P. Heins, Renee J. Sifri, Dillon T. Gentekos, Rachael E. Coleman, Peter T. Wolczanski*, Thomas R. Cundari, Brett P. Fors, Kyle M. Lancaster, and Samantha N. MacMillan
Inorg. Chem. 2021, 60, 18662.
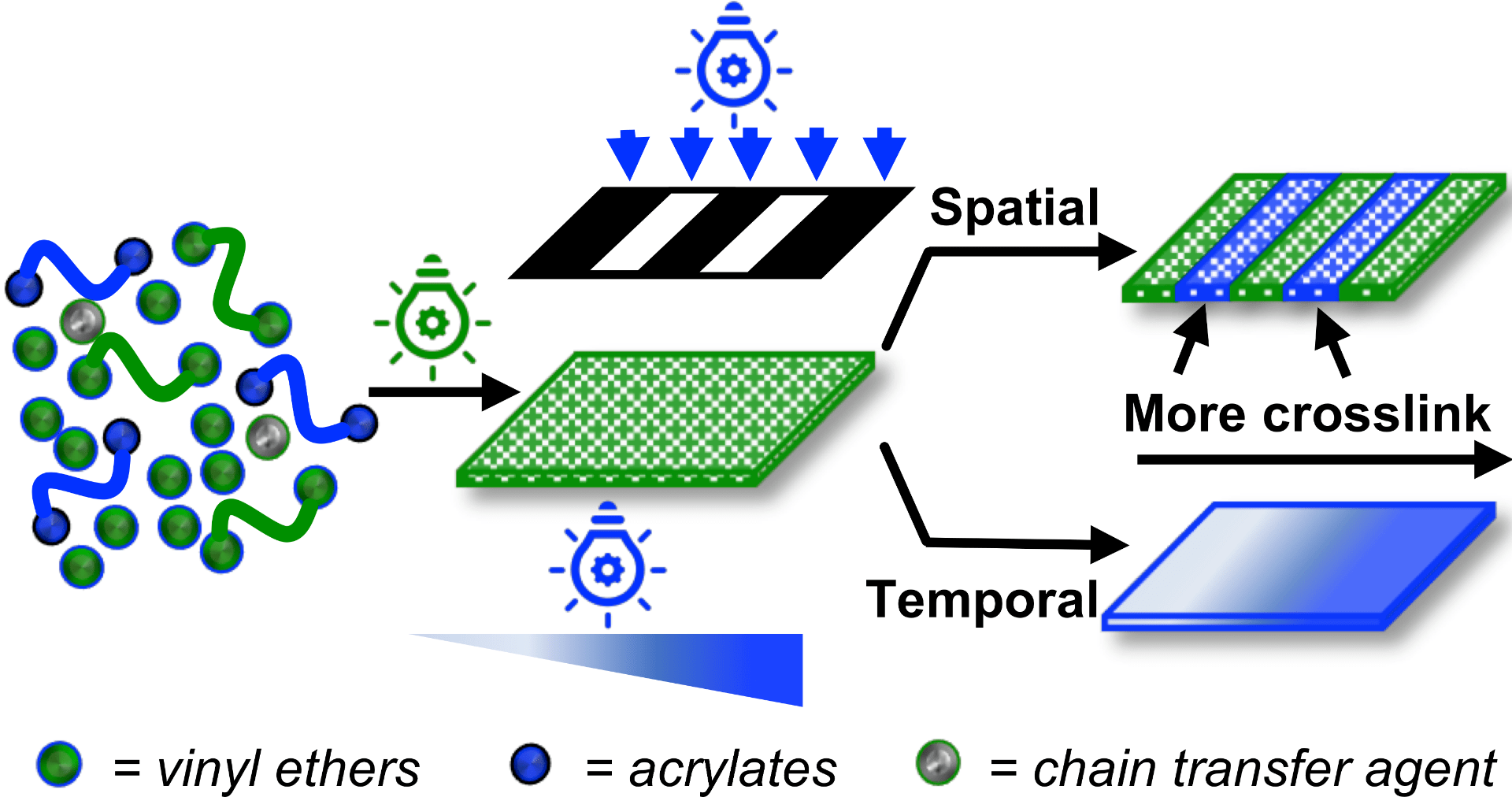
71) Photoswitching Cationic and Radical Polymerizations: Spatiotemporal Control of Thermoset Properties
Yuting Ma, Veronika Kottisch, Elizabeth A. McLoughlin, Zachary W. Rouse, Michael J. Supej, Shefford P. Baker, and Brett P. Fors
J. Am. Chem. Soc. 2021, 143, 21200.
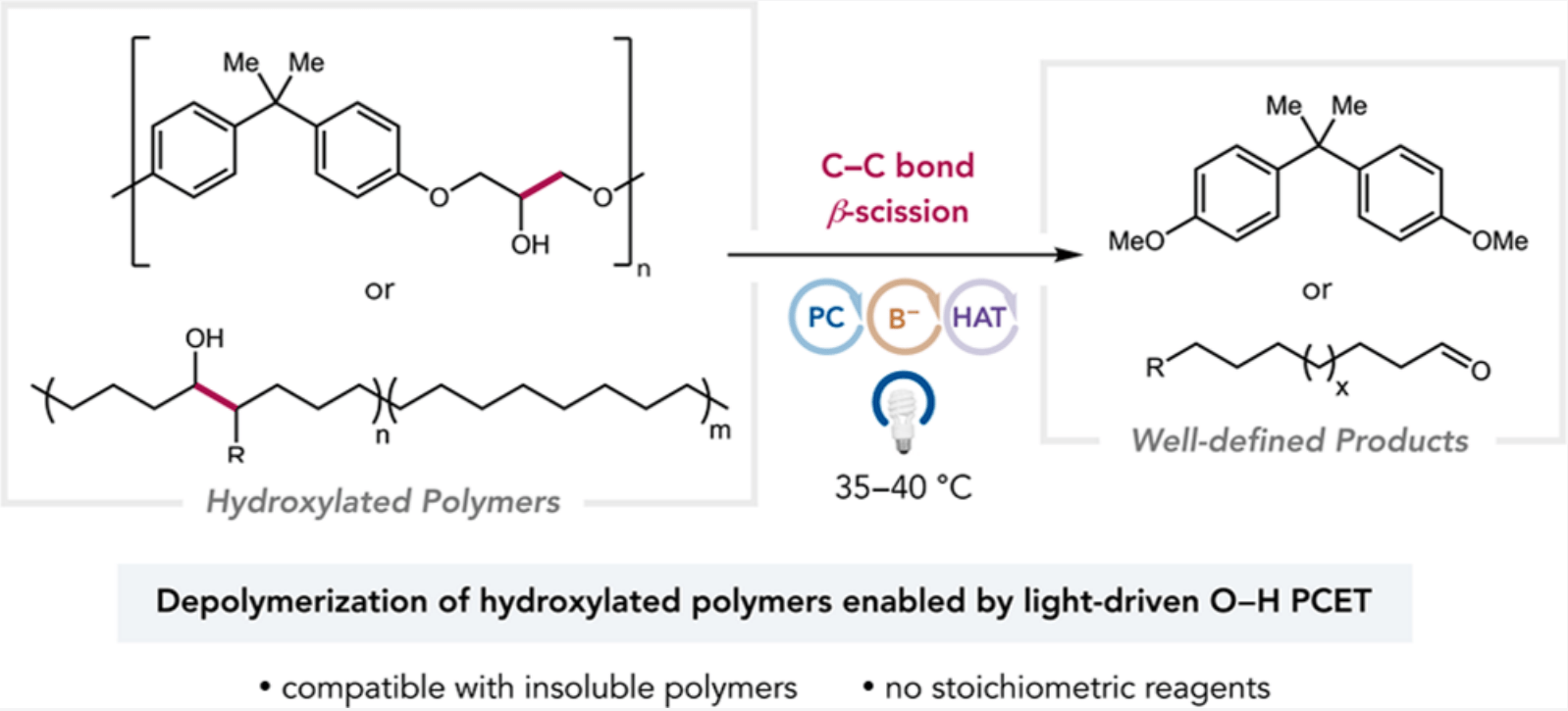
70) Depolymerization of Hydroxylated Polymers via Light-Driven C-C Bond Cleavage
Suong T. Nguyen, Elizabeth A. McLoughlin,‡ James H. Cox,‡ Brett P. Fors, and Robert R. Knowles
J. Am. Chem. Soc. 2021, 143, 12268.
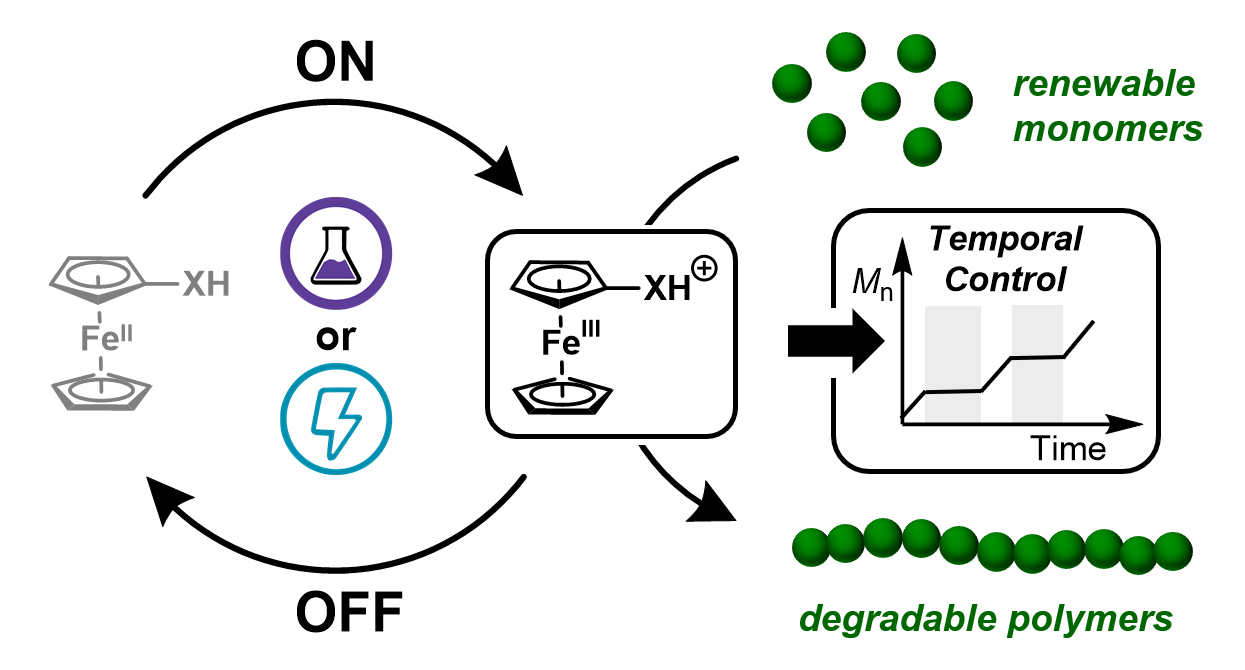
69) Reversible Redox Controlled Acids for Cationic Ring-Opening Polymerization
Michael J. Supej, Elizabeth A. McLoughlin, Jesse H. Hsu, and Brett P. Fors
Chem. Sci. 2021, 12, 10544.
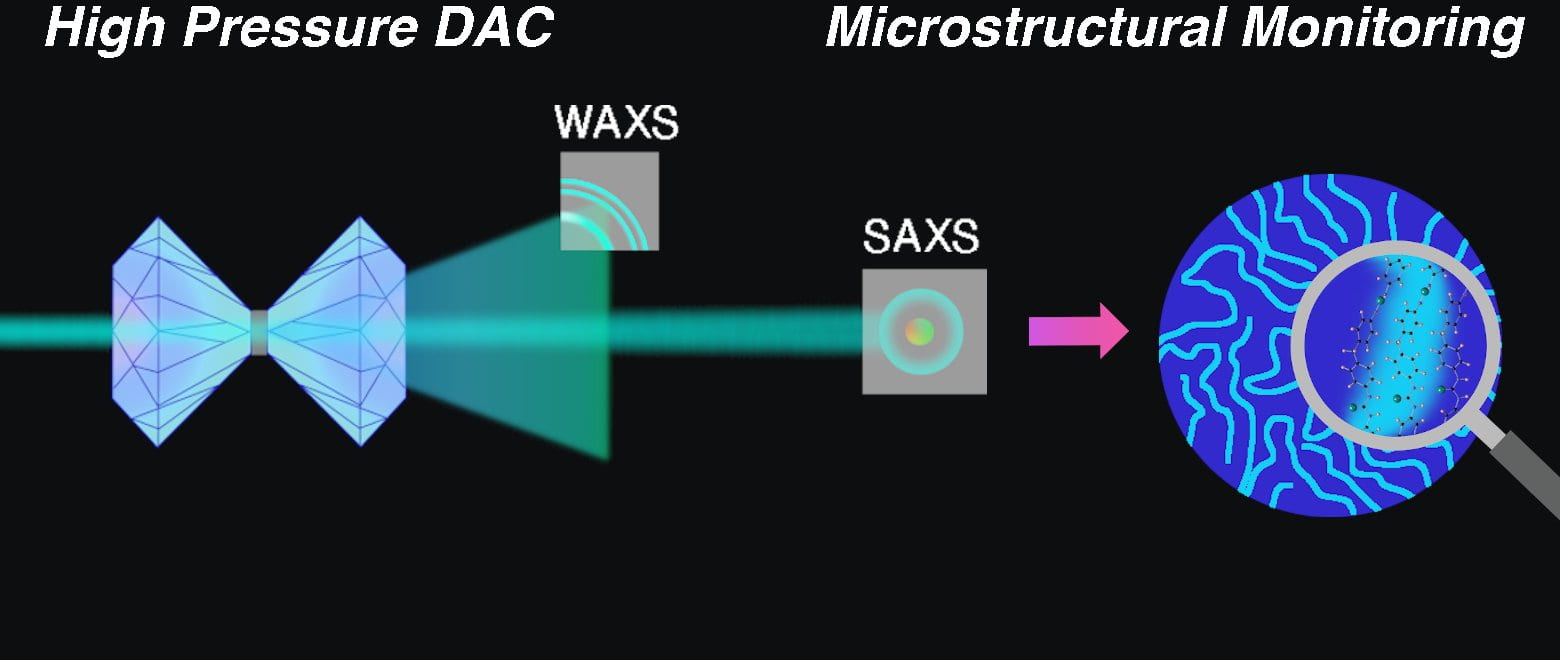
68) Microstructural Evolution of Polyurea Under Hydrostatic Pressure
Stephanie I. Rosenbloom, Steven J. Yang, Nikolas J. Tsakeredes, Brett P. Fors, and Meredith N. Silberstein
Polymer, 2021, 227, 123845.
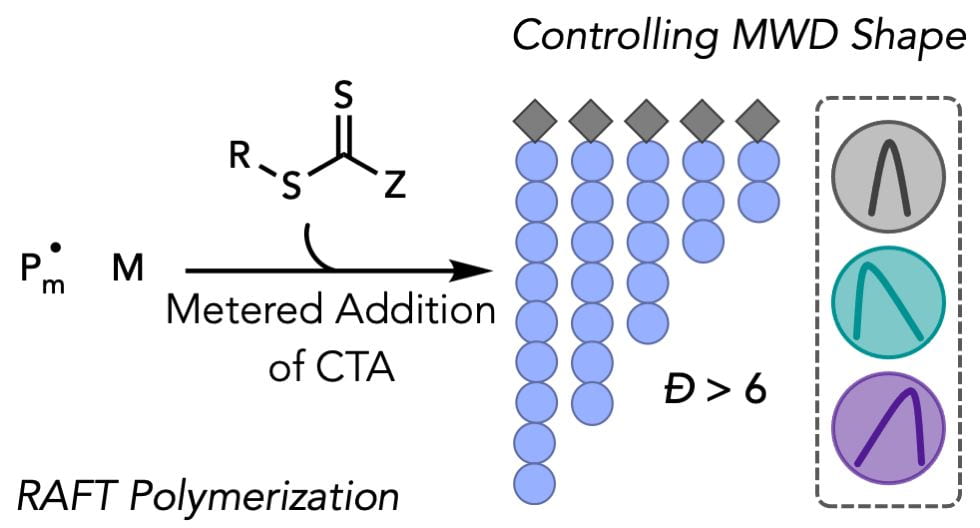
67) Achieving Molecular Weight Distribution Shape Control and Broad Dispersities using RAFT Polymerization
Stephanie I. Rosenbloom, Renee J. Sifri, and Brett P. Fors
Polym. Chem. 2021, 12, 4910.

66) Effect of Structural Ordering on the Charge Storage Mechanism of p-Type Organic Electrode Materials
Brian M. Peterson,‡ Cara N. Gannett,‡ Luis Melecio-Zambrano, Brett P. Fors, and Héctor D. Abruña
ACS Appl. Mater. Interfaces, 2021, 13, 7135.
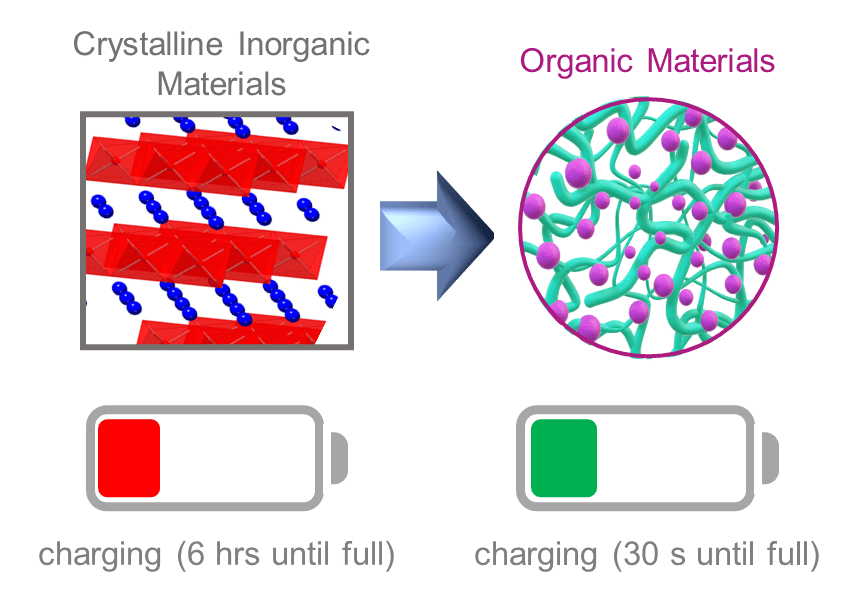
65) Organic Electrode Materials for Fast-Rate, High-Power Battery Applications
Cara N. Gannett, Luis Melecio-Zambrano,‡ Monica Theibault,‡ Brian M. Peterson, Brett P. Fors, and Héctor D. Abruña
Materials Reports: Energy, 2021, 1, 100008.
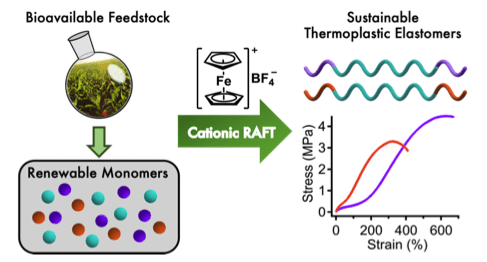
64) Sustainable Thermoplastic Elastomers Produced via Cationic RAFT Polymerization
Scott W. Spring, Red Smith-Sweetser, Stephanie I. Rosenbloom, Renee J. Sifri and Brett P. Fors
Polym. Chem. 2021, 12, 1097.
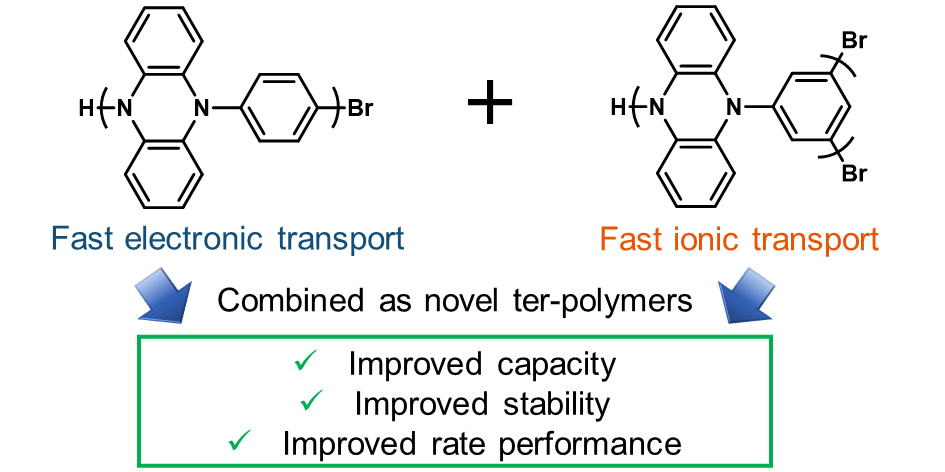
63) Performance Optimization and Fast Rate Capabilities of Novel Polymer Cathode Materials through Balanced Electronic and Ionic Transport
Cara Gannett, Brian M. Peterson, Luis Melecio-Zambrano, Colleen Q. Trainor, Brett P. Fors and Héctor D. Abruña
J. Mater. Chem. A, 2021, 9, 5657.
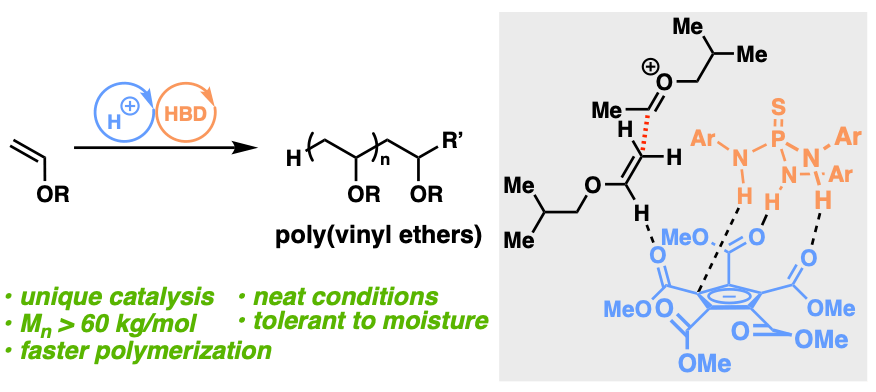
62) Hydrogen Bond Donor‐Catalyzed Cationic Polymerization of Vinyl Ethers
Veronika Kottisch, Janis Jermaks, Joe-Yee Mak, Ryan Woltornist, Tristan Lambert, and Brett P. Fors
Angew. Chem. Int. Ed. 2020, 59, 4535.
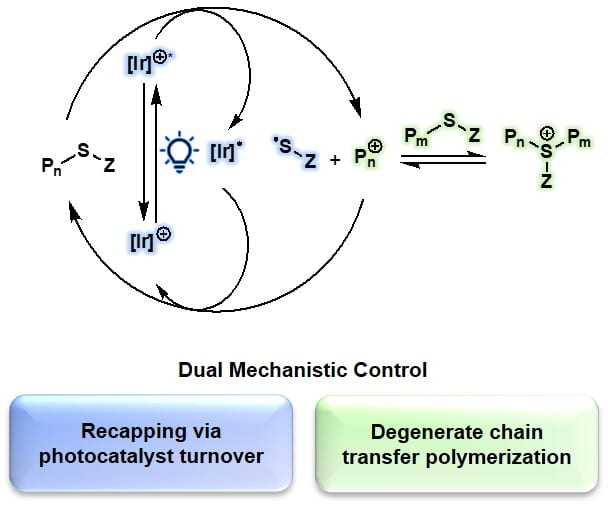
61) Photocontrolled Cationic Degenerate Chain Transfer Polymerizations via Thioacetal Initiators
Renee J. Sifri, Audrey J. Kennedy, and Brett P. Fors
Polym. Chem. 2020, 11, 6499.
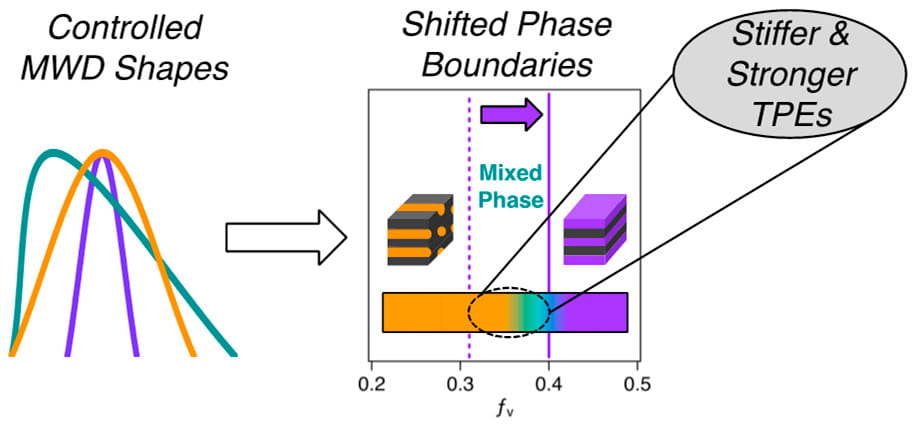
60) Shifting Boundaries: Controlling Molecular Weight Distribution Shape for Mechanically Enhanced Thermoplastic Elastomers
Stephanie I. Rosenbloom and Brett P. Fors
Macromolecules, 2020, 53, 7479.
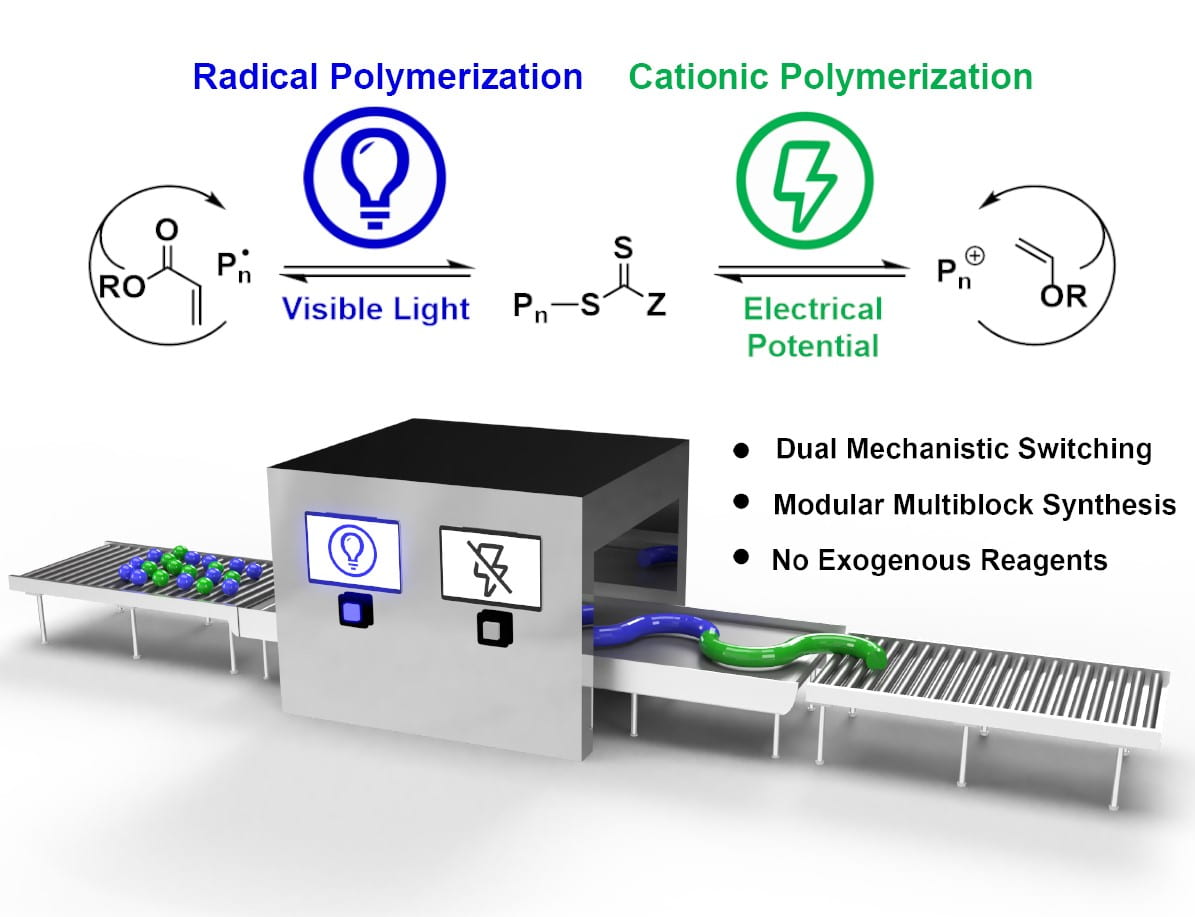
59) Dual Stimuli Switching: Interconverting Cationic and Radical Mechanisms with Electricity and Light
Michael J. Supej, Brian M. Peterson, and Brett P. Fors
Chem, 2020, 6, 1508.
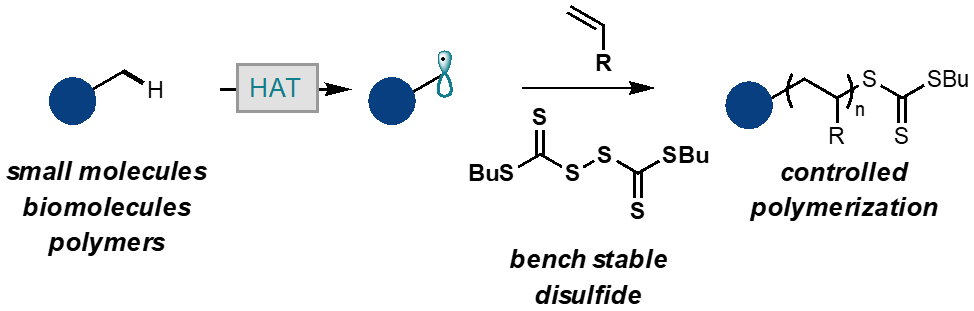
58) Photocontrolled Radical Polymerization from Hydridic C-H Bonds
Erin E. Stache, Veronika Kottisch, and Brett P. Fors
J. Am. Chem. Soc. 2020, 142, 4581.
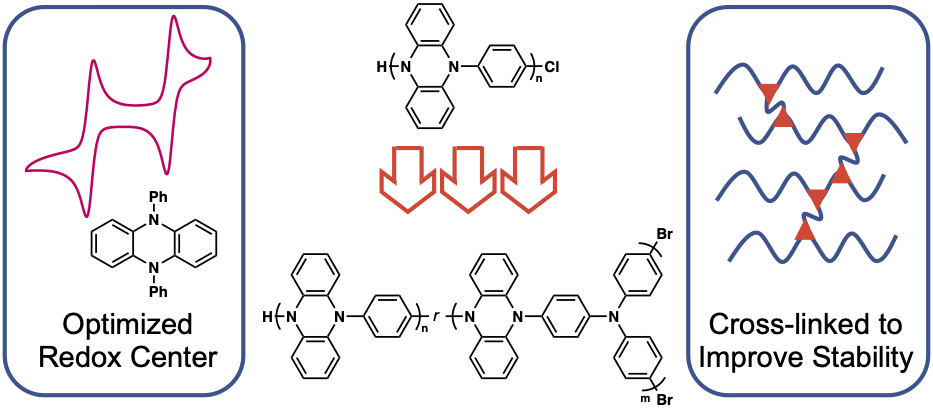
57) Cross-linking Effects on Performance Metrics of Phenazine-Based Polymer Cathodes
Cara N. Gannett,‡ Brian M. Peterson,‡ Luxi Shen, Jeesoo Seok, Brett P. Fors, and Héctor Abruña
ChemSusChem, 2020, 13, 2428.
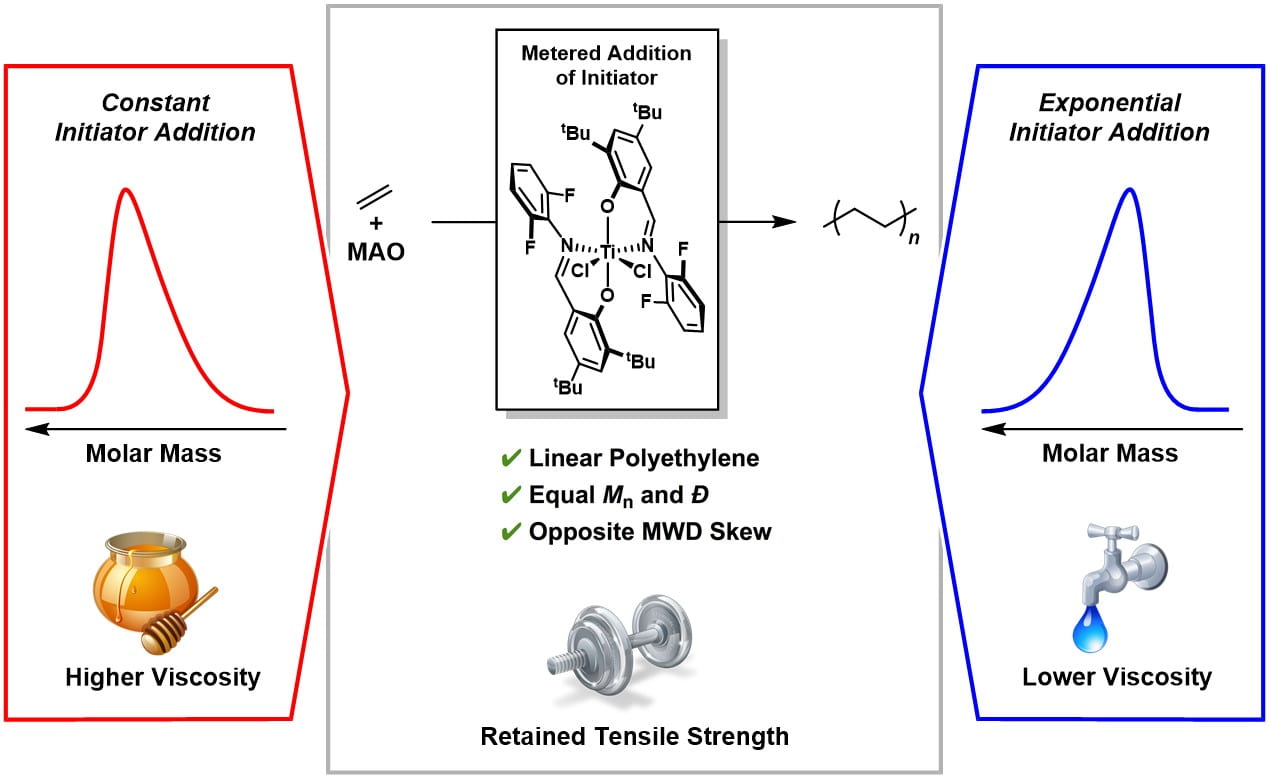
56) Controlling the Shape of Molecular Weight Distributions in Coordination Polymerization and Its Impact on Physical Properties
Renee J. Sifri,‡ Omar Padilla-Vélez,‡ Geoffrey W. Coates , and Brett P. Fors
J. Am. Chem. Soc. 2020, 142, 1443.

55) Tailor-Made Thermoplastic Elastomers: Customisable Materials via Modulation of Molecular Weight Distributions
Stephanie I. Rosenbloom, Dillon T. Gentekos, Meredith N. Silberstein, and Brett P. Fors
Chem. Sci. 2020, 11, 1361.
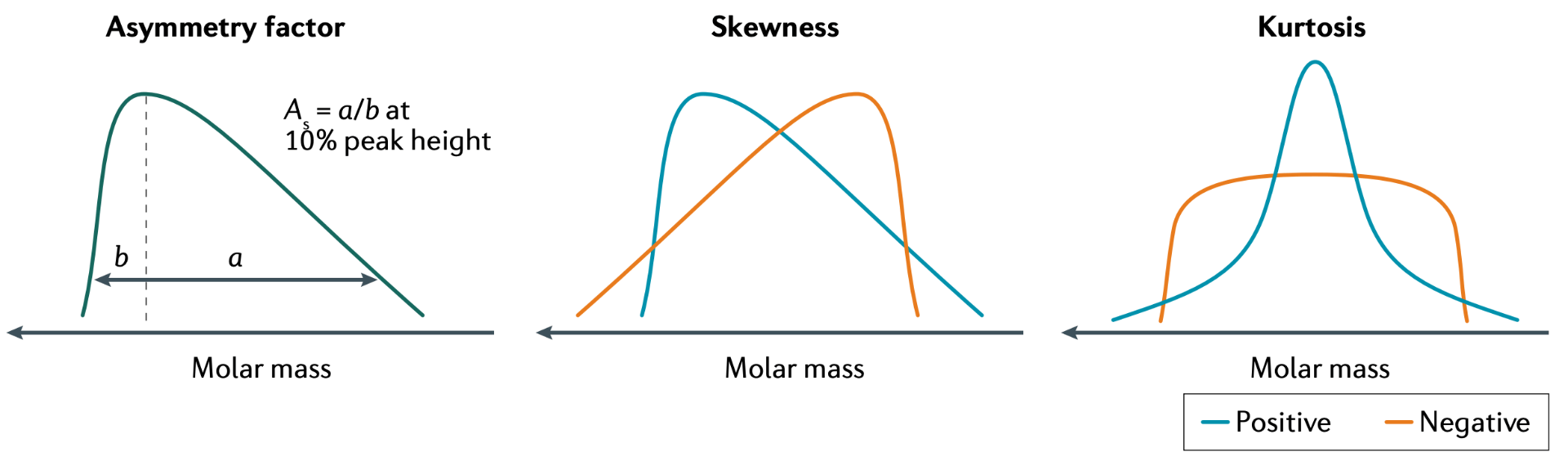
54) Controlling Polymer Properties Through the Shape of the Molecular-Weight Distribution
Dillon T. Gentekos, Renee J. Sifri, and Brett P. Fors
Nat. Rev. Mater. 2019, 4, 761.
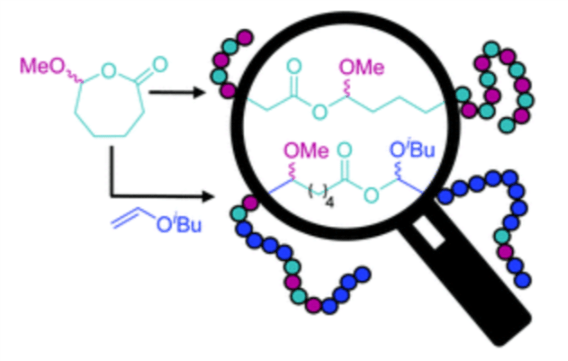
53) Hydrolytically-Degradable Homo- and Copolymers of a Strained Exocyclic Hemiacetal Ester
Angelika E. Neitzel, Leonel Barreda, Jacob T. Trotta, Grant W. Fahnhorst, Thomas J. Haversang, Thomas R. Hoye, Brett P. Fors, and Marc A. Hillmyer
Polym. Chem. 2019, 10, 4573.
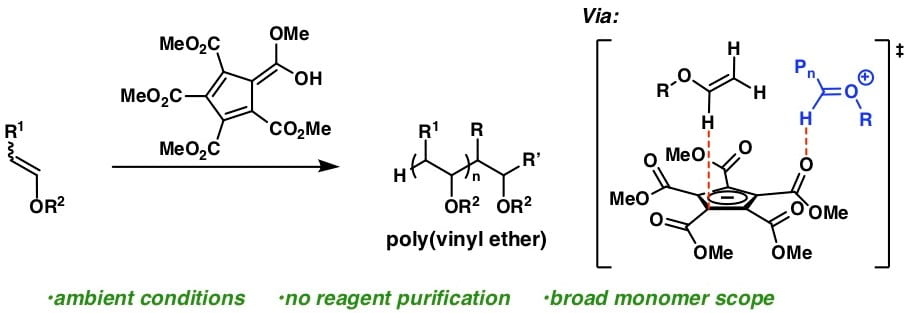
52) Controlled Cationic Polymerization: Single-Component Initiation Under Ambient Conditions
Veronika Kottisch, Jacob O’Leary, Quentin Michaudel, Erin E. Stache, Tristan H. Lambert, and Brett P. Fors
J. Am. Chem. Soc. 2019, 141, 10605.
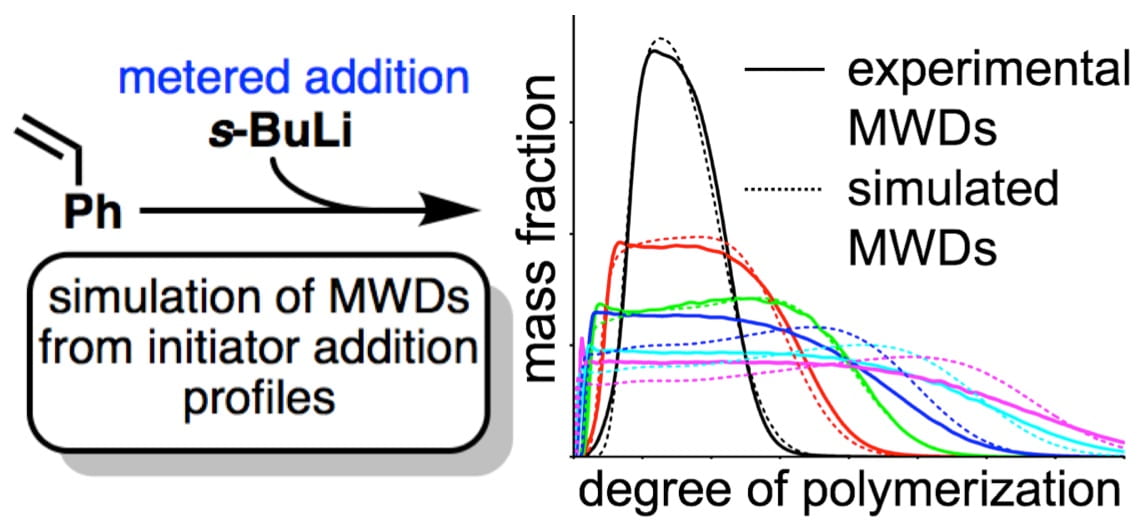
51) Predictive Design of Polymer Molecular Weight Distributions in Anionic Polymerization
Sergii Domanskyi, Dillon Gentekos, Vladimir Privman, and Brett P. Fors
Polym. Chem. 2020, 11, 326.
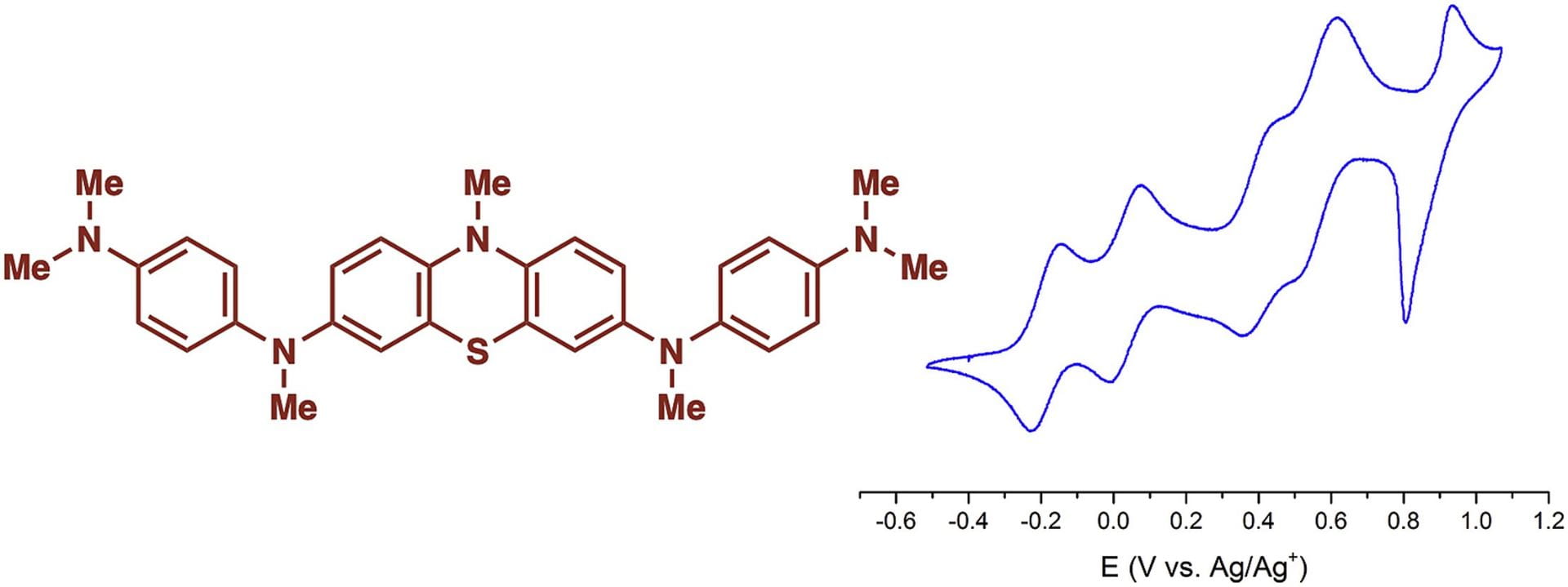
50) Elucidation of the Electrochemical Behavior of Phenothiazine-Based Polyaromatic Amines
Brian M. Peterson, Luxi Shen, Gerickson J. Lopez, Cara N. Gannett, Dong Ren, Héctor D. Abruña, and Brett P. Fors
Tetrahedron, 2019, 75, 4244.
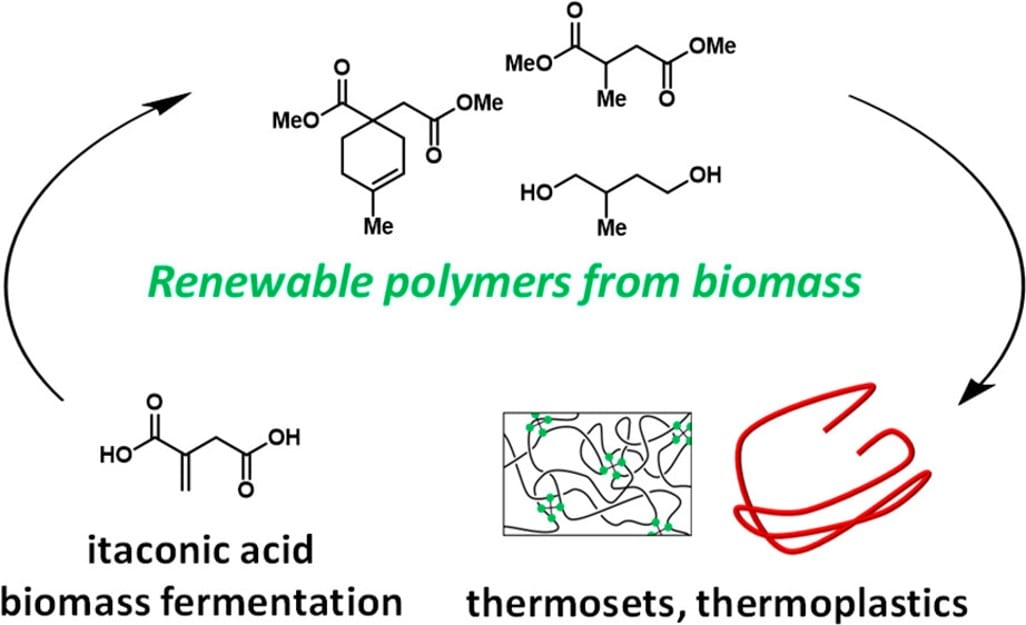
49) Renewable Thermosets and Thermoplastics from Itaconic Acid
Jacob T. Trotta,‡ Annabelle Watts,‡ Allison R. Wong, Anne M. LaPointe, Marc A. Hillmyer, and Brett P. Fors
ACS Sustainable Chem. Eng. 2019, 7, 2691.
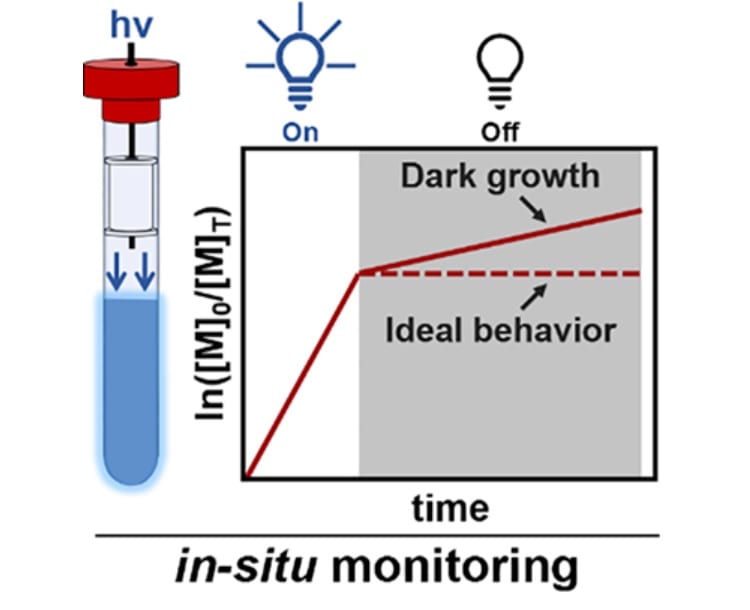
48) What Happens in the Dark? Assessing the Temporal Control of Photo-Mediated Controlled Radical Polymerizations
Neil D. Dolinski, Zachariah A. Page, Emre H. Discekici, David Meis, In-Hwan Lee, Glen R. Jones, Richard Whitfield, Xiangcheng Pan, Blaine G. McCarthy, Sivaprakash Shanmugam, Veronika Kottisch, Brett P. Fors, Cyrille Boyer, Garret M. Miyake, Krzysztof Matyjaszewski, David M. Haddleton, Javier Read de Alaniz, Athina Anastasaski, and Craig J. Hawker
Journal of Polymer Science Part A: Polymer Chemistry, 2019, 57, 268.

47) On Demand Switching of Polymerization Mechanism and Monomer Selectivity with Orthogonal Stimuli
Brian M. Peterson, Veronika Kottisch, Michael J. Supej, and Brett P. Fors
ACS Cent. Sci. 2018, 4, 1228.
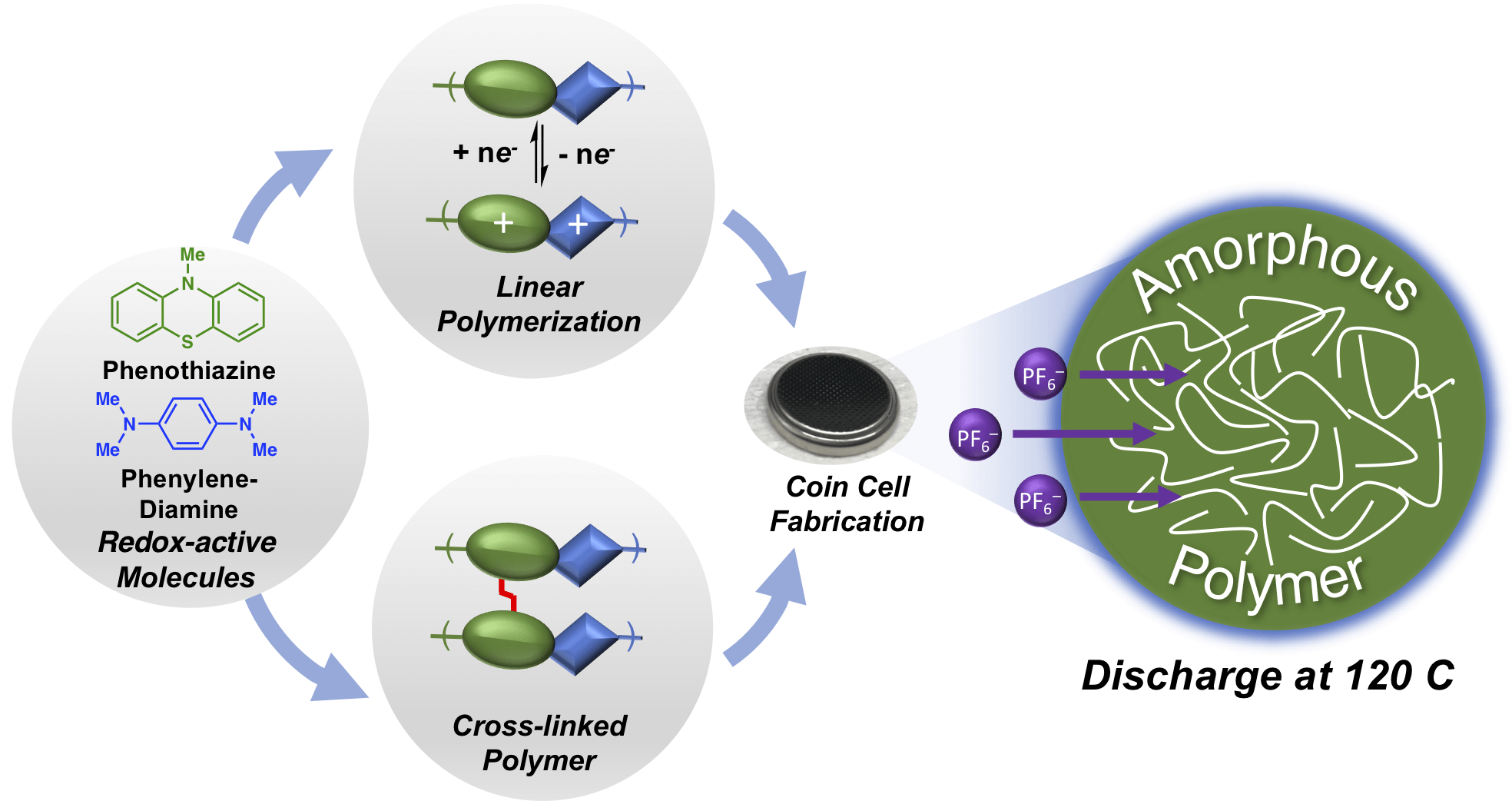
46) Phenothiazine-Based Polymer Cathode Materials with Ultra-High Power Densities for Lithium Ion Batteries
Brian M. Peterson,‡ Dong Ren,‡ Luxi Shen,‡ You-Chi Mason Wu, Burak Ulgut, Geoffrey W. Coates, Héctor D. Abruña, and Brett P. Fors
ACS Appl. Energy Mater. 2018, 1, 3560.
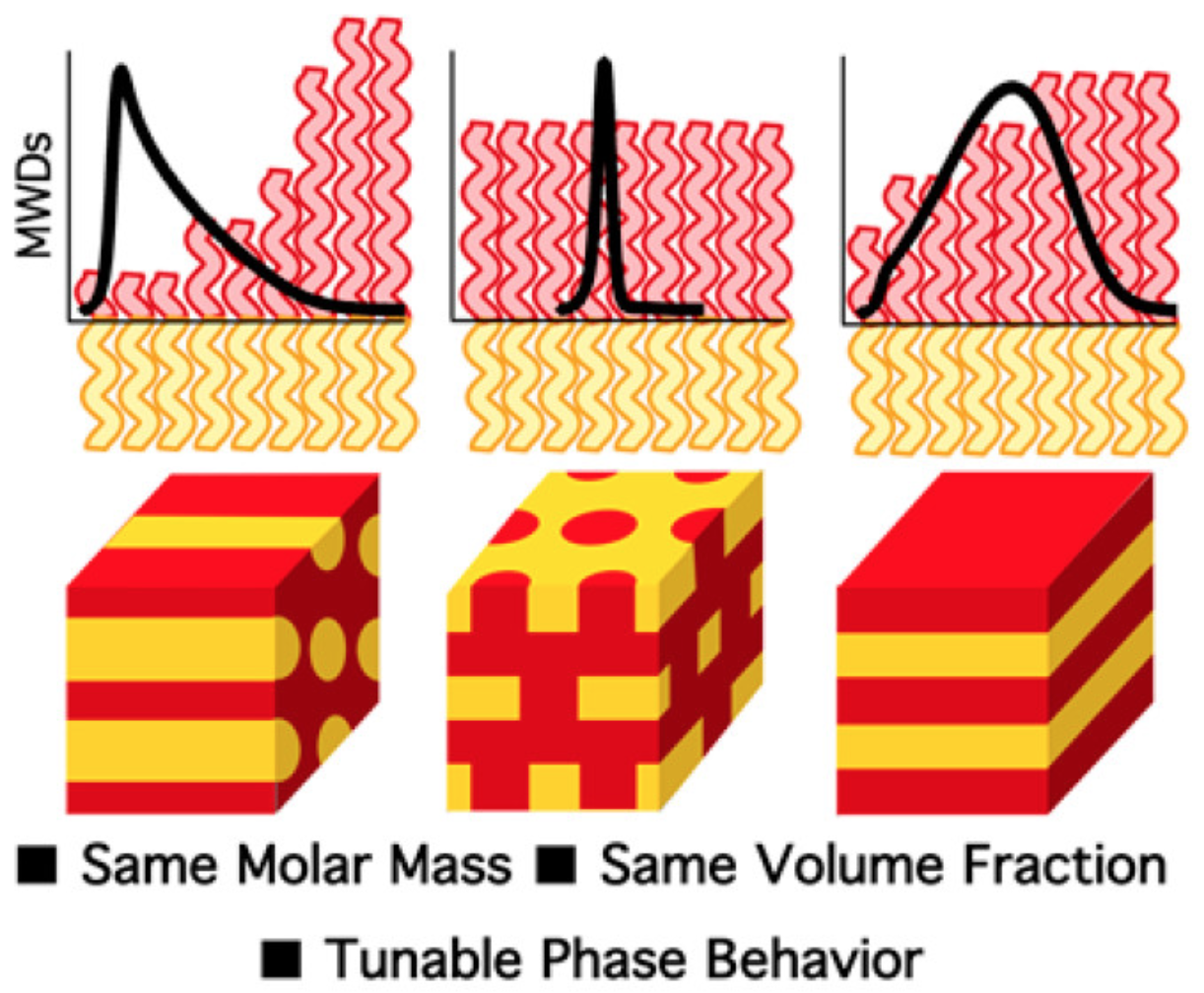
45) Molecular Weight Distribution Shape as a Versatile Approach to Tailoring Block Copolymer Phase Behavior
Dillon T. Gentekos and Brett P. Fors
ACS Macro Lett. 2018, 7, 677.
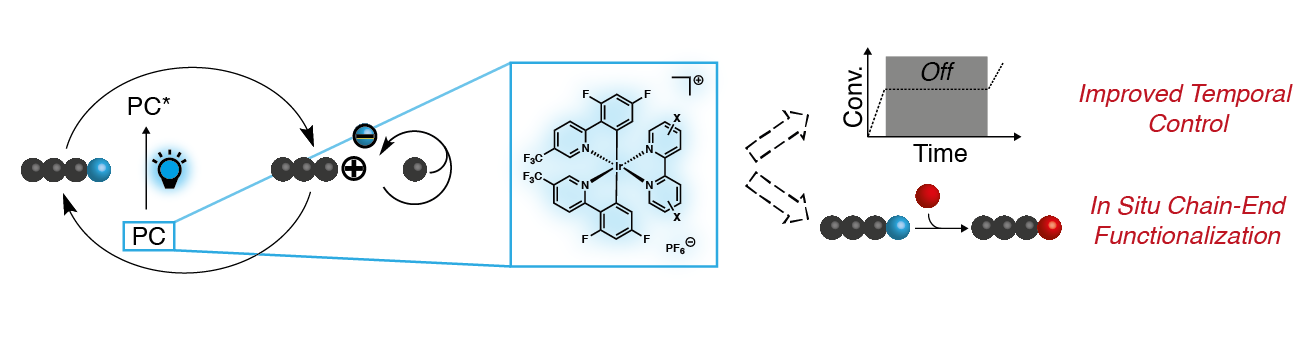
44) Enhancing Temporal Control and Enabling Chain-End Modification in Photoregulated Cationic Polymerizations by Using Ir-Based Catalysts
Veronika Kottisch, Michael J. Supej, and Brett P. Fors
Angew. Chem. Int. Ed. 2018, 57, 8260.
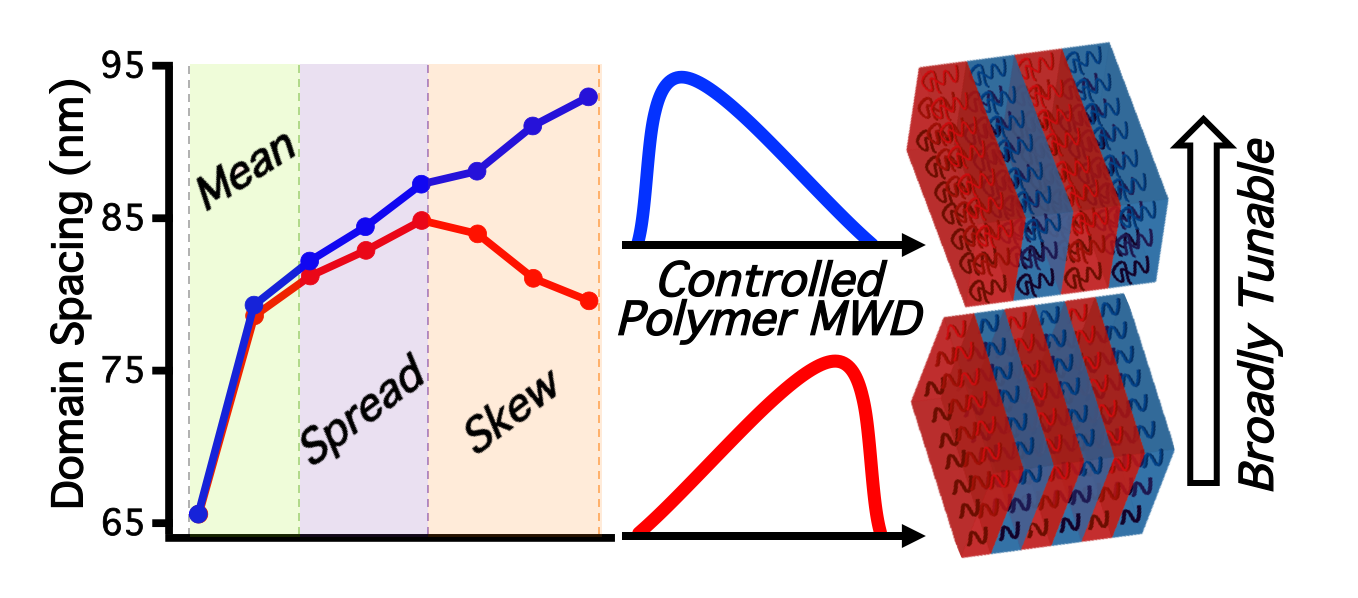
43) Exploiting Molecular Weight Distribution Shape to Tune Domain Spacing in Block Copolymer Thin Films
Dillon T. Gentekos,‡ Junteng Jia,‡ Erika S. Tirado, Katherine P. Barteau, Detlef-M Smilgies, Robert A. Distasio Jr., and Brett P. Fors
J. Am. Chem. Soc. 2018, 140, 4639.
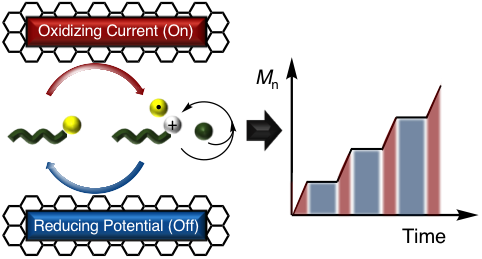
42) Electrochemically Controlled Cationic Polymerization of Vinyl Ethers
Brian M. Peterson, Song Lin, and Brett P. Fors
J. Am. Chem. Soc. 2018, 140, 2076.
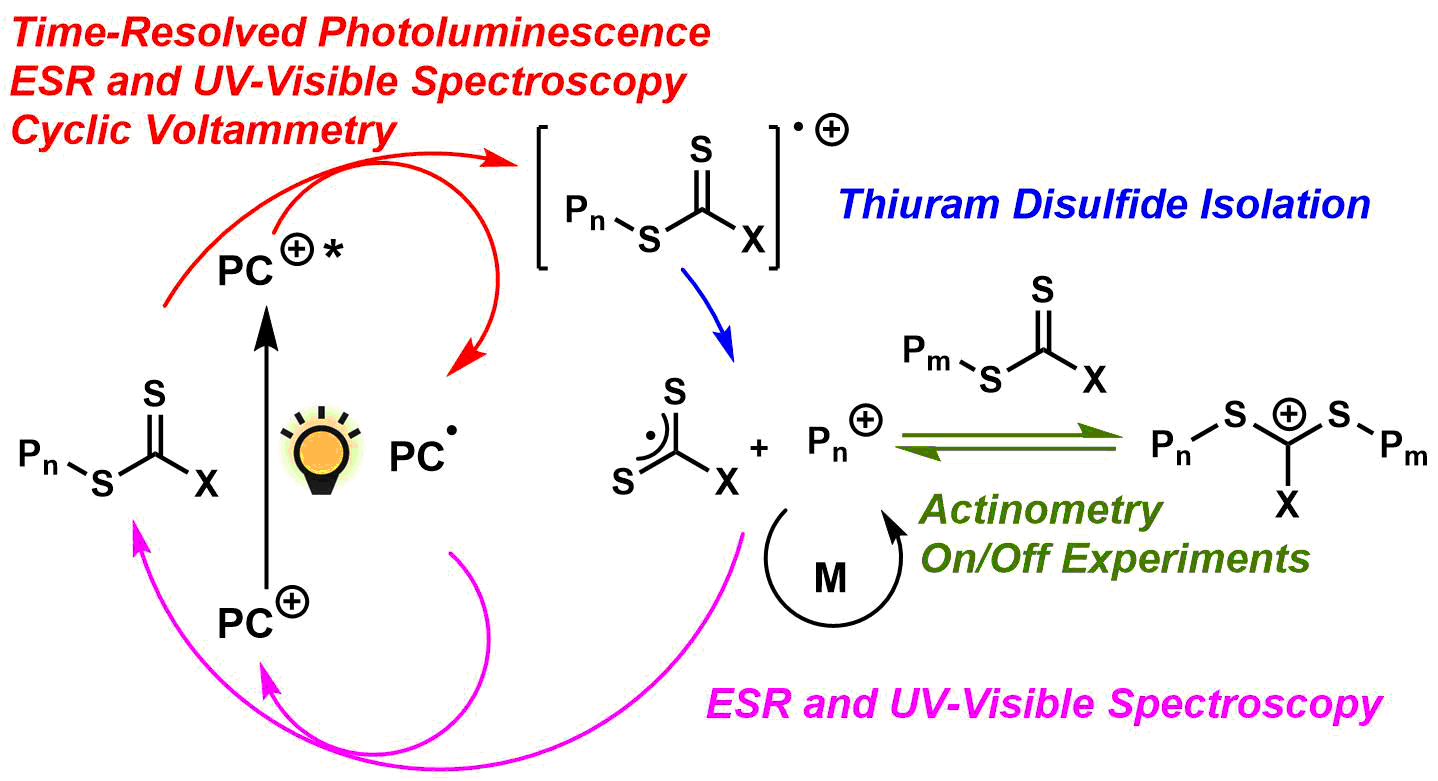
41) Mechanistic Insight into the Photocontrolled Cationic Polymerization of Vinyl Ethers
Quentin Michaudel, Timothee Chauvire, Veronika Kottisch, Michael J. Supej, Katherine J. Stawiasz, Luxi Shen, Warren R. Zipfel, Héctor D. Abruña, Jack H. Freed, and Brett P. Fors
J. Am. Chem. Soc. 2017, 139, 15530.
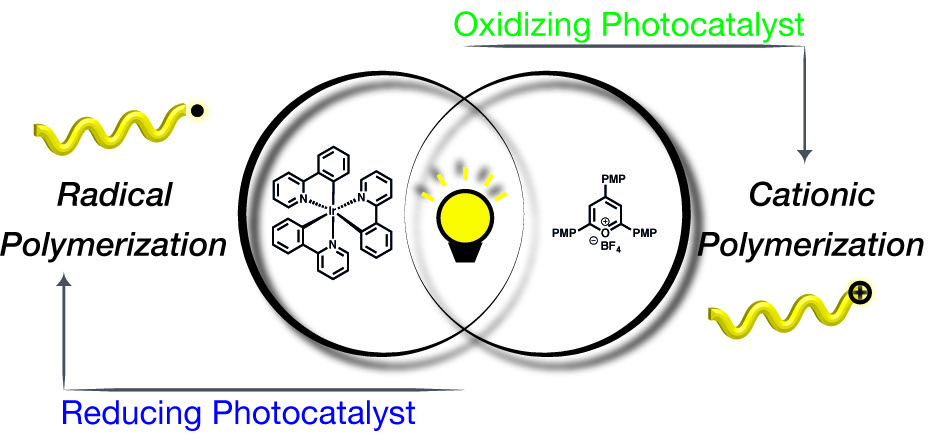
40) Photocontrolled Interconversion of Cationic and Radical Polymerizations
Veronika Kottisch, Quentin Michaudel, and Brett P. Fors
J. Am. Chem. Soc. 2017, 139, 10665.
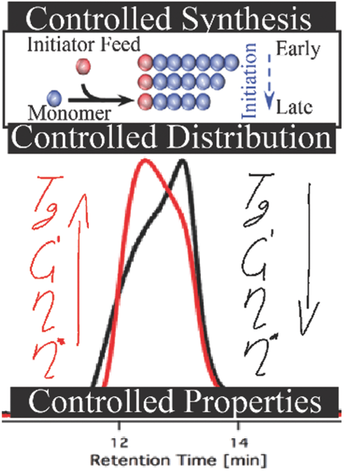
39) Manipulation of Molecular Weight Distribution Shape as a New Strategy to Control Processing Parameters
Milena Nadgorny, Dillon T. Gentekos, Zeyun Xiao, S. Parker Singleton, Brett P. Fors, and Luke A. Connal
Macromol. Rapid Commun. 2017, 1700352.
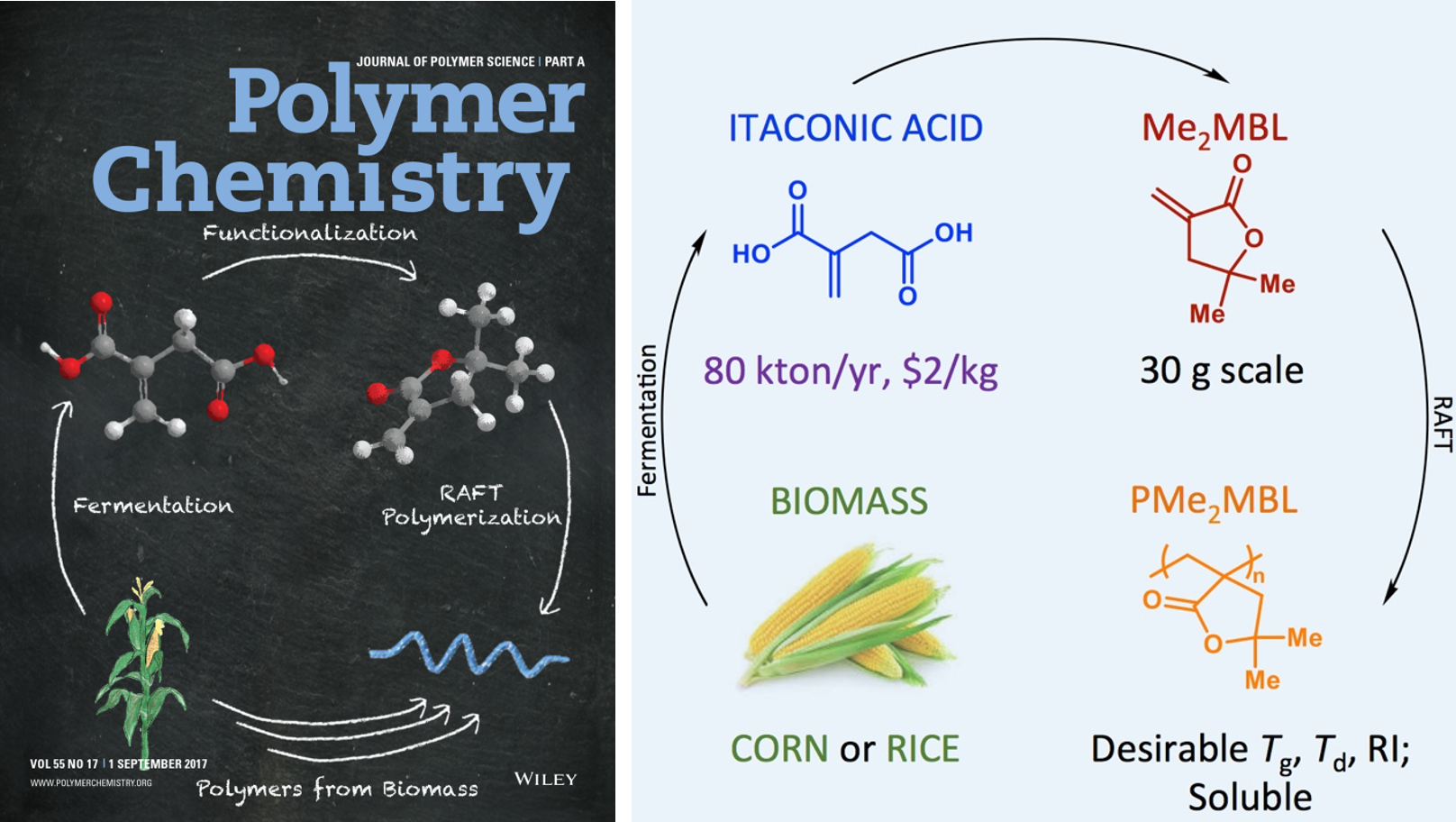
38) Synthesis of Methylene Butyrolactone Polymers from Itaconic Acid
Jacob T. Trotta, Mengjuan Jin, Katherine J. Stawiasz, Quentin Michaudel, Wei-Liang Chen, and Brett P. Fors
J. Polym. Sci. A Polym. Chem. 2017, 55, 2730.
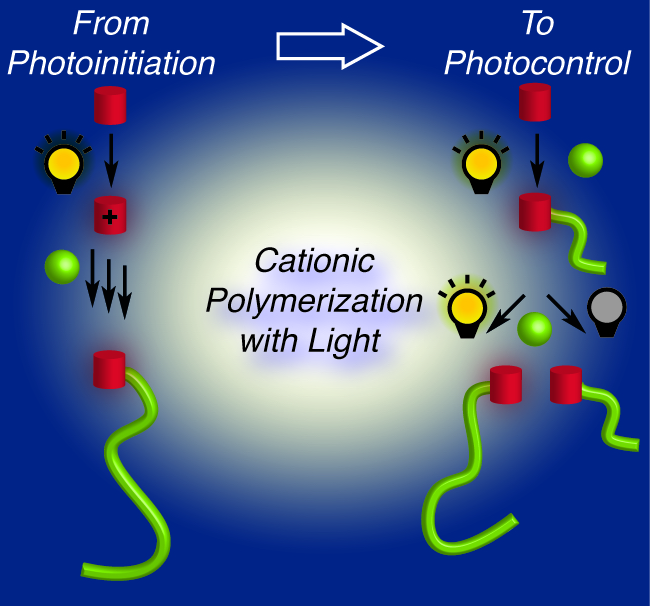
37) Cationic Polymerization: From Photoinitiation to Photocontrol
Quentin Michaudel, Veronika Kottisch, and Brett P. Fors
Angew. Chem. Int. Ed. 2017, 56, 9670.
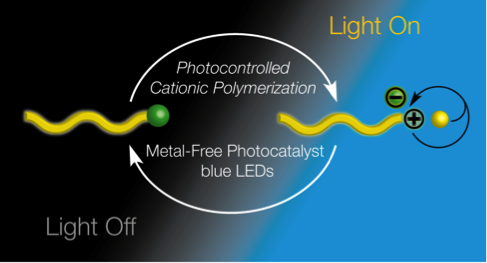
36) Cationic Polymerization of Vinyl Ethers Controlled by Visible Light
Veronika Kottisch,‡ Quentin Michaudel,‡ and Brett P. Fors
J. Am. Chem. Soc. 2016, 138, 15535.

35) Storing Information at the Molecular Level: Efficient Synthesis of "Barcode" Polymers, Preview
Quentin Michaudel and Brett P. Fors
Chem, 2016, 1, 23.
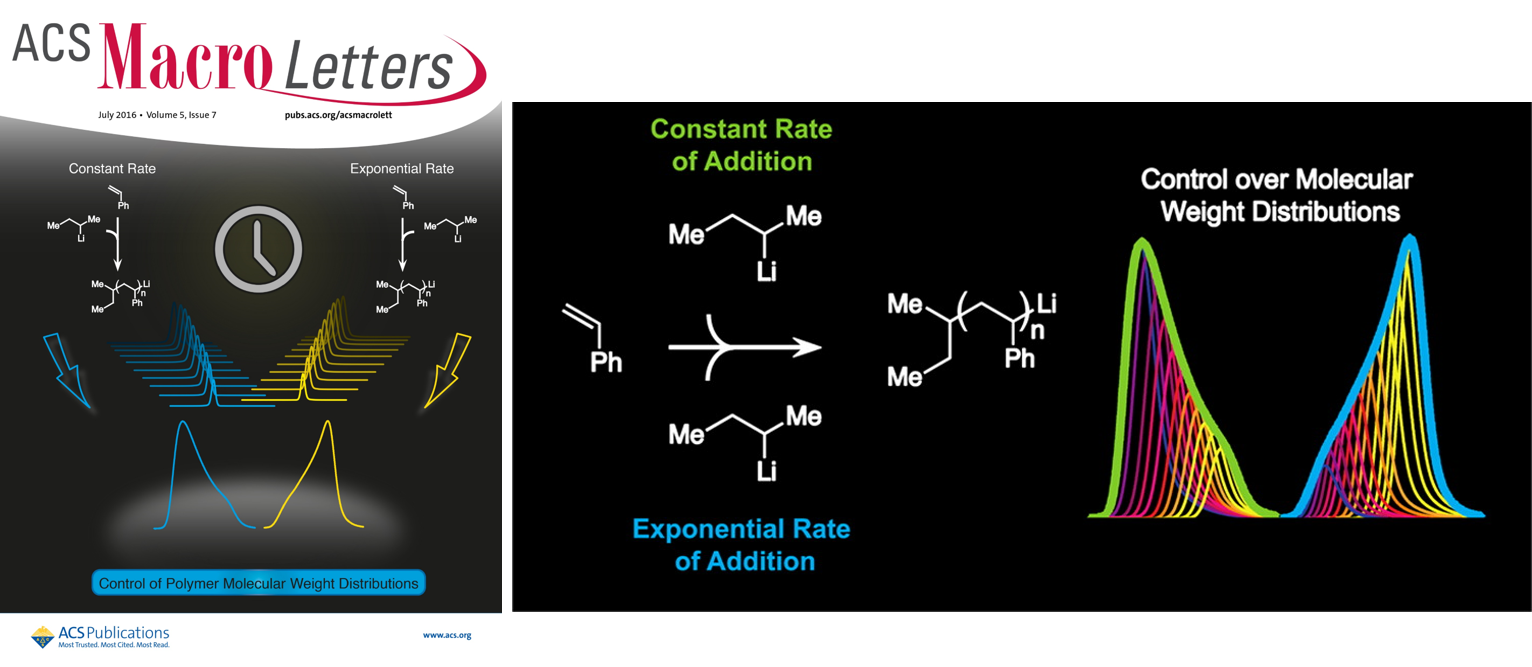
34) Shaping the Future of Molecular Weight Distributions in Anionic Polymerizations
Veronika Kottisch, Dillion T. Gentekos, and Brett P. Fors
ACS Macro Lett. 2016, 5, 796.
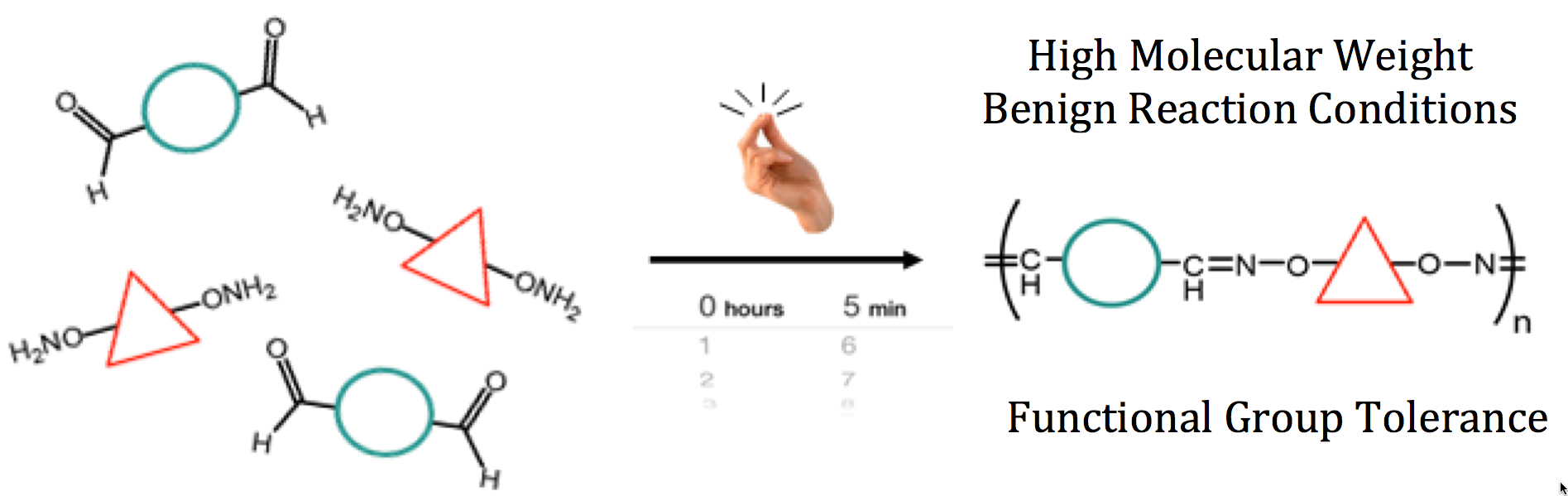
33) Extremely Rapid and Versatile Synthesis of High Molecular Weight Step Growth Polymers via Oxime Click Chemistry
Joe Collins, Zeyun Xiao, Andrea Espinosa-Gomez, Brett P. Fors, and Luke A. Connal
Polym. Chem. 2016, 7, 2581.
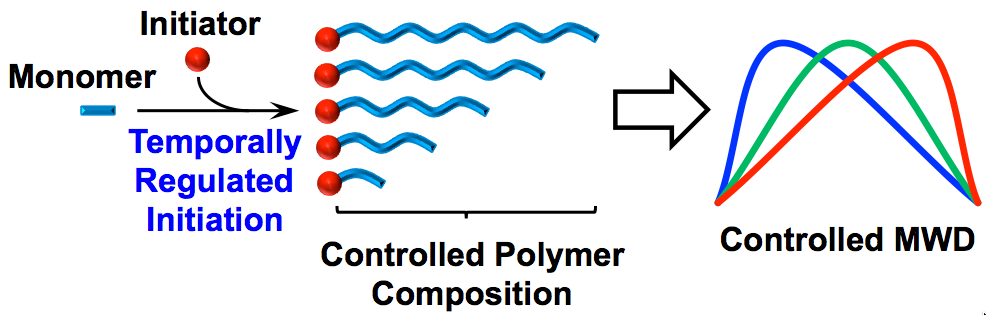
32) Beyond Dispersity: Deterministic Control of Polymer Molecular Weight Distributions
Dillon T. Gentekos, Lauren N. Dupuis, and Brett P. Fors
J. Am. Chem. Soc. 2016, 138, 1848.
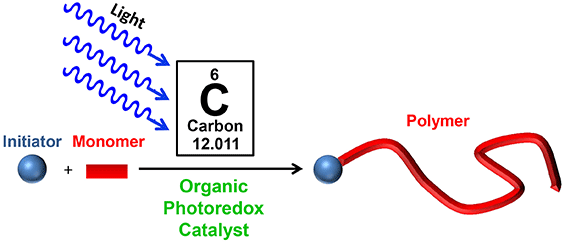
31) Organic Catalysts for Photocontrolled Polymerizations
Jacob T. Trotta and Brett P. Fors
Synlett, 2016, 27, 702.
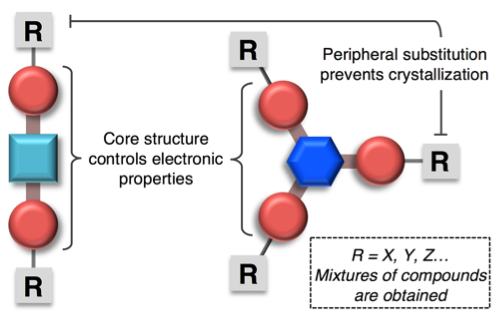
30) Synthesis of Amorphous Monomeric Glass Mixtures of Organic Electronic Applications
You-Chi Mason Wu, Michel F. Molaire, Catherine R. DeBlase, David S. Weiss, and Brett P. Fors
J. Org. Chem. 2015, 80, 12740.
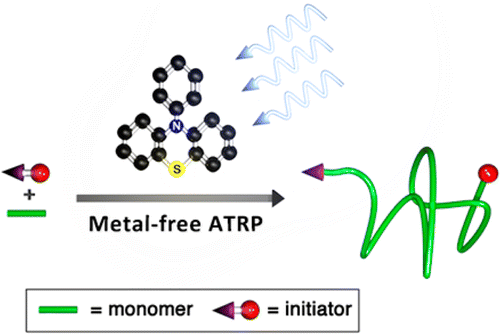
29) Metal-Free Atom Transfer Radical Polymerization
Nicolas J. Treat, Hazel Sprafke, John W. Kramer, Paul G. Clark, Bryan E. Barton, Javier Read de Alaniz, Brett P. Fors, and Craig J. Hawker
J. Am. Chem. Soc. 2014, 136, 16096.
Prior to Cornell
28) Hannah P. Luehmann, Lisa Detering, Brett P. Fors, Eric D. Pressly, Pamela K. Woodard, Gwendalyn J. Randolph, Robert J. Gropler, Craig J. Hawker, Yongjian Liu “PET/CT Imaging of Chemokine Receptors in Inflammatory Atherosclerosis Using Targeted Nanoparticles” J. Nucl. Med. 2016, DOI: 10.2967/jnumed.115.166751.
27) Anna Melker, Brett P. Fors, Craig J. Hawker, Justin E. Poelma “Continuous Flow Synthesis of Poly(methyl methacrylate) via a Light-Mediated Controlled Radical Polymerization” J. Polym. Sci., Part A: Polym. Chem. 2015, 53, 2693.
26) Nicolas J. Treat, Brett P. Fors, John W. Kramer, Matthew Christianson, Chien-Yang Chiu, Javier Read de Alaniz, Craig J. Hawker “Controlled Radical Polymerization of Acrylates Regulated by Visible Light” ACS Macro Lett. 2014, 3, 580.
25) Maxwell J. Robb, Brandon Newton, Brett P. Fors, Craig J. Hawker “One-Step Synthesis of Unsymmetrical N-Alkyl-N’-Aryl Perylene Diimides” J. Org. Chem. 2014, 79, 6360.
24) Lauren M. Misch, Alexander Birkel, C. Adrian Figg, Brett P. Fors, Craig J. Hawker, Galen D. Stucky, Ram Seshadri “Rapid Microwave-Assisted Sol-Gel Preparation of Pd-Substituted LnFeO3 (Ln = Y, La): Phase Formation and Catalytic Activity” Dalton Trans. 2014, 43, 2079.
23) Brett P. Fors, Justin E. Poelma, Matthew S. Menyo, Maxwell J. Robb, Daniel M. Spokoyny, John W. Kramer, J. Herbert Waite, Craig J. Hawker “Fabrication of Unique Chemical Patterns and Surface Concentration Gradients with Visible Light” J. Am. Chem. Soc. 2013, 135, 14106.
22) Justin E. Poelma, Brett P. Fors, Gregory F. Meyers, John W. Kramer, Craig J. Hawker “Fabrication of Complex 3-Dimensional Polymer Brush Nanostructures via Light-Mediated Living Radical Polymerization” Angew. Chem. Int. Ed. 2013, 52, 6844.
21) Frank A. Leibfarth, Kaila M. Mattson, Brett P. Fors, Hazel A. Collins, Craig J. Hawker “External Regulation of Controlled Polymerizations” Angew. Chem. Int. Ed. 2013, 52, 199.
20) Brett P. Fors, Craig J. Hawker “Control of a Living Radical Polymerization of Methacrylates by Light” Angew. Chem. Int. Ed. 2012, 51, 8850.
19) Ekaterina V. Vinogradova, Nathaniel H. Park, Brett P. Fors, Stephen L. Buchwald “Palladium-Catalyzed Synthesis of N-Aryl Carbamates” Org. Lett. 2013, 15, 1394.
18) Ekaterina V. Vinogradova, Brett P. Fors, Stephen L. Buchwald “Palladium-Catalyzed Cross-Coupling of Aryl Chlorides and Triflates with Sodium Cyanate: A Practical Synthesis of Unsymmetrical Ureas” J. Am. Chem. Soc. 2012, 134, 11132.
17) Satoshi Ueda, Siraj Ali, Brett P. Fors, Stephen L. Buchwald “Me3(OMe)tBuXPhos: A Surrogate Ligand for Me4tBuXPhos in Palladium-Catalyzed C–N and C–O Bond-Forming Reactions” J. Org. Chem. 2012, 77, 2543.
16) Simon Breitler, Nathan J. Oldenhuis, Brett P. Fors, Stephen L. Buchwald “Synthesis of Unsymmetrical Diarylureas via Pd-Catalyzed C–N Cross-Coupling Reactions” Org. Lett. 2011, 13, 3262.
15) Brett P. Fors and Stephen L. Buchwald “Commentary on ‘A New, Efficient and Recyclable Lanthanum(III) Oxide-Catalyzed C-N Cross-Coupling’ by S. Narayana Murthy, B. Madhav, V. Prakash Reddy, and Y. V. D. Nageswar, Adv. Synth. Catal. 2010, 352, 3241–3245” Adv. Synth. Catal. 2010, 352, 3119.
14) Brett P. Fors, Stephen L. Buchwald “A Multiligand Based Pd Catalyst for C–N Cross-Coupling Reactions” J. Am. Chem. Soc. 2010, 132, 15914.
13) Xiaoxing Wu, Brett P. Fors, Stephen L. Buchwald “A Single Phosphine Ligand Allows Palladium-Catalyzed Intermolecular C–O Bond Formation with Secondary and Primary Alcohols” Angew. Chem. Int. Ed. 2011, 50, 9943.
12) Debrata Maiti, Brett P. Fors, Jaclyn L. Henderson, Yoshinori Nakamura, Stephen L. Buchwald “Palladium-Catalyzed Coupling of Functionalized Primary and Secondary Amines with Aryl and Heteroaryl Halides: Two Ligands Suffice in Most Cases” Chem. Sci. 2011, 2, 57.
11) Karin Dooleweerdt, Brett P. Fors, Stephen L. Buchwald “Pd-Catalyzed Cross-Coupling of Amides and Aryl Mesylates” Org. Lett. 2010, 12, 2350.
10) Brett P. Fors, Stephen L. Buchwald “Pd-Catalyzed Conversion of Aryl Chlorides, Triflates, and Nonaflates to Nitroaromatics” J. Am. Chem. Soc. 2009, 131, 12898.
9) John R. Naber, Brett P. Fors, Xiaoxing Wu, Jonathon T. Gunn, Stephen L. Buchwald “Stille Cross-Coupling Reactions of Aryl Mesylates and Tosylates Using a Biarylphosphine Based Catalyst System” Heterocycles 2010, 80, 1215.
8) Brijesh Bhayana, Brett P. Fors, Stephen L. Buchwald “A Versatile Catalyst System for Suzuki-Miyaura Cross-Coupling Reactions of C(sp2)-Tosylates and Mesylates” Org. Lett. 2009, 11, 3954.
7) Brett P. Fors, Karin Dooleweerdt, Qingle Zeng, Stephen L. Buchwald “An Efficient System for the Pd-Catalyzed Cross-Coupling of Amides and Aryl Chlorides” Tetrahedron 2009, 65, 6576.
6) Brett P. Fors, Nicole R. Davis, Stephen L. Buchwald “An Efficient Process for Pd-Catalyzed C–N Cross-Coupling Reactions of Aryl Iodides: Insight Into Controlling Factors” J. Am. Chem. Soc. 2009, 131, 5766.
5) Brett P. Fors, Donald A. Watson, Mark R. Biscoe, Stephen L. Buchwald “A Highly Active Catalyst for Pd-Catalyzed Amination Reactions: Cross-Coupling Reactions Using Aryl Mesylates and the Highly Selective Monoarylation of Primary Amines Using Aryl Chlorides” J. Am. Chem. Soc. 2008, 130, 13552.
4) Brett P. Fors, Philip Krattiger, Eric Strieter, Stephen L. Buchwald “Water-Mediated Catalyst Preactivation: An Efficient Protocol for C–N Cross-Coupling Reactions” Org. Lett. 2008, 10, 3505.
3) Mark R. Biscoe, Brett P. Fors, Stephen L. Buchwald “A New Class of Easily Activated Palladium Precatalysts for Facile C–N Cross-Coupling Reactions and the Low Temperature Oxidative Addition of Aryl Chlorides” J. Am. Chem. Soc. 2008, 130, 6686.
2) Ryan A. Altman, Brett P. Fors, Stephen L. Buchwald “Pd-Catalyzed Amination Reactions of Aryl Halides Using Bulky Biarylmonophosphine Ligands” Nature Protocols 2007, 2, 2881.
1) Harry R. Chobanian, Brett P. Fors, Linus S. Lin “A Facile Microwave-Assisted Palladium-Catalyzed Cyanation of Aryl Chlorides” Tetrahedron Lett. 2006, 47, 3303.
